Sorry, nothing in cart.
The global pharmaceutical market is valued at about USD 1.6 trillion, as per IQVIA estimates. Generic medicines form a large share of this market and lead in volume across many countries.
India is the source of nearly 20% of the world’s generic medicines, which are exported to over 200 countries.

Global buyers are sourcing more from India. This shift is due to strong quality standards, cost benefits, and reliable manufacturing.
After COVID-19, many countries shifted supply chains to reduce risk. India became a trusted partner by providing a stable supply, skilled talent, and a broad export reach. Many buyers worldwide obtain specialty generics from India.
Understanding Regulatory Frameworks in Destination Markets
Every country has rules to ensure that medicines are safe, effective, and of high quality.
Below, we explain key global regulators and their approval systems.
Major Global Regulatory Authorities
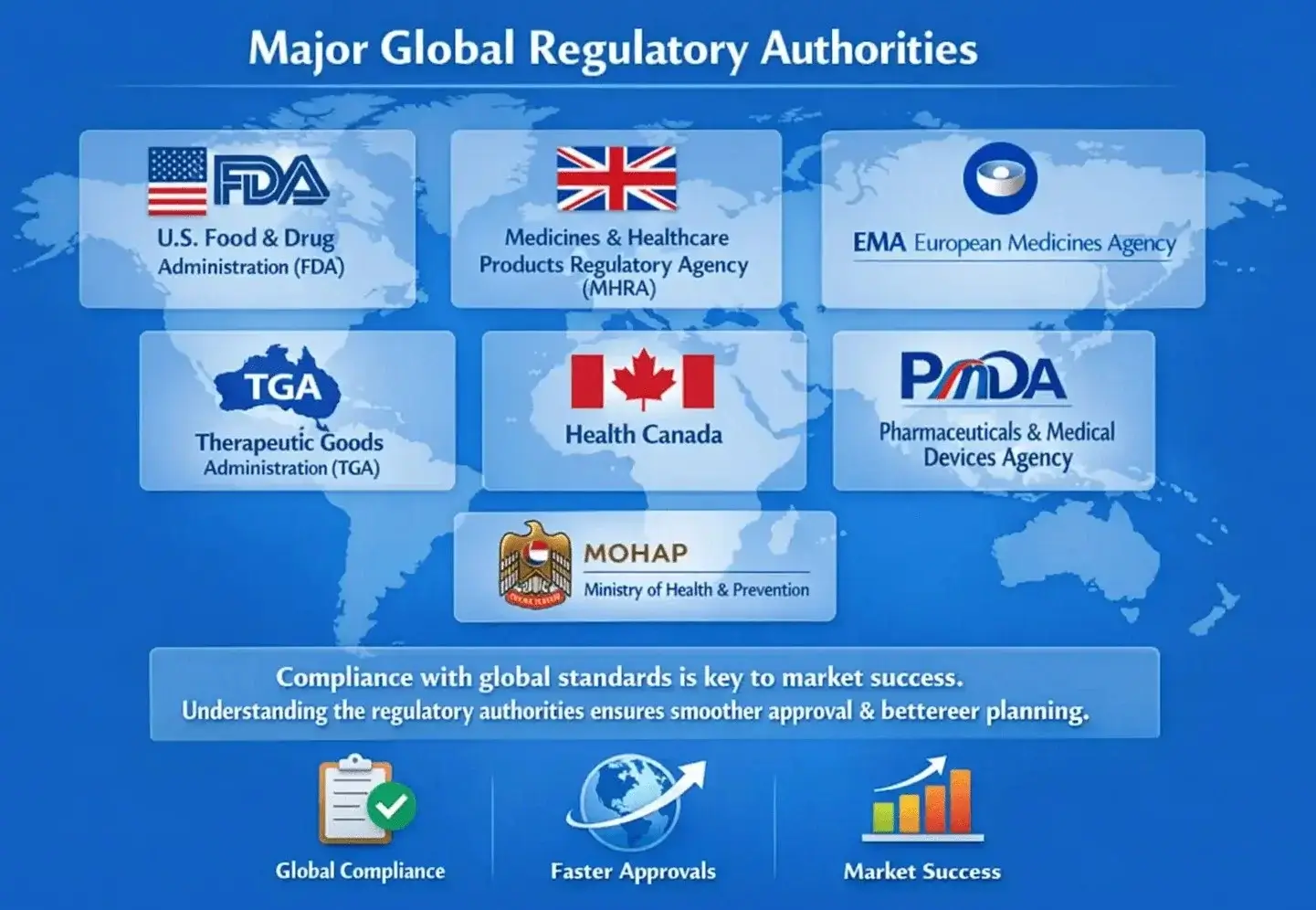
Every country has a team that approves medicines, and each team follows its own rules. To ship to other countries, exporters need to know the major global players that regulate this process.
Compliance with global standards is crucial for exporters. It not only earns them trust but also speeds up approval processes, leading to better market success in the long run. Exporters can plan more effectively and avoid issues by understanding the regulatory bodies.
Key Compliance Requirements
Pharma exporters face tough rules before they can enter global markets. Here are the key regulatory documents and quality standards that companies have to follow.
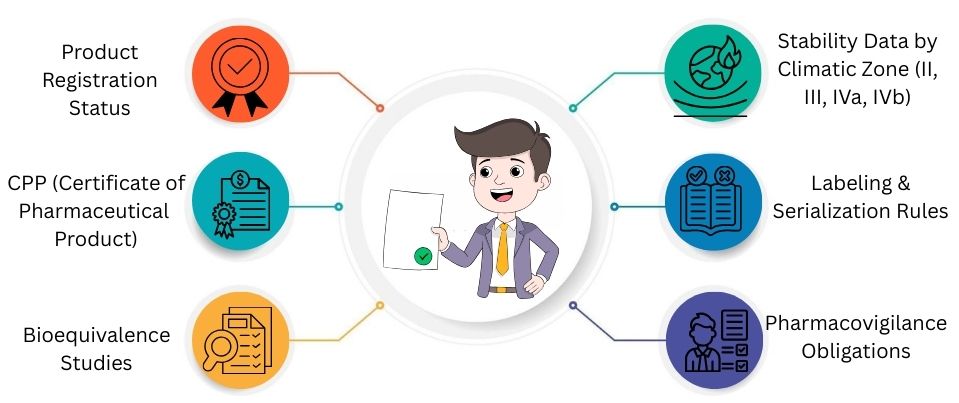
Product Registration Status
The product must get approval or registration in the exporting country. This is needed before it can enter new markets.
CPP (Certificate of Pharmaceutical Product)
A CPP checks that the product meets quality standards. It also ensures the product is officially authorized in its country.
Bioequivalence Studies
Bioequivalence studies show that a generic medicine is just as effective as the brand-name version.
Stability Data by Climatic Zone (II, III, IVa, IVb)
Stability studies show that the medicine is safe. It remains effective in different conditions, like various temperatures and humidity levels.
Labeling & Serialization Rules
Labels must follow each country’s rules. This includes content, language, barcodes, safety warnings, and tracking systems.
Pharmacovigilance Obligations
Companies need to monitor and report any side effects to the authorities, even after their products hit the shelves. This means tracking and documenting these issues and sending them to regulators.
Deep Dive into Technical Documentation (Highly Important Section)
Technical documents prove a medicine is safe, effective, and of high quality. Below are the key regulatory files and why they matter for buyers and regulators.
CTD (Common Technical Document) Format
The CTD is a global format for submitting drug data to regulators.
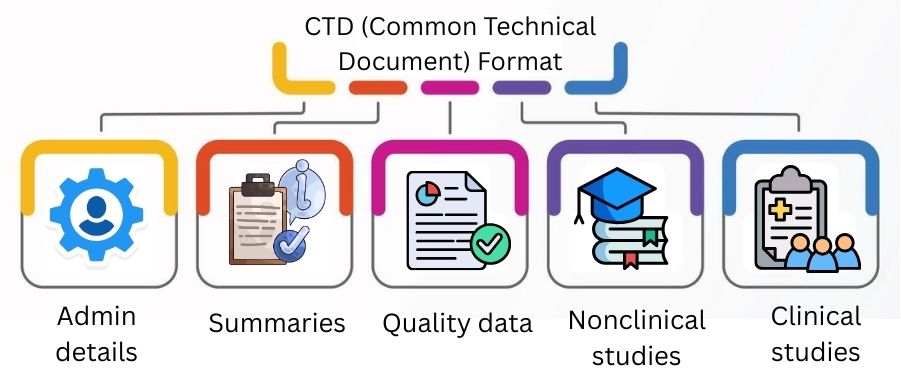
It has five modules:
It helps with faster review and approval.
Batch-Specific COA vs Master COA
A Master COA lists standard test limits and product specifications. A batch-specific COA presents the actual test results for each lot. It confirms that every batch meets approved quality standards.
Process Validation Reports
These reports demonstrate that the manufacturing process operates in a stable, repeatable manner. They include batch data, test results, and conclusions. This shows that the process reliably makes safe, quality products.
Cleaning Validation Reports
Cleaning validation reports show that the equipment is cleaned well after production. They ensure that residues, detergents, and microbes are cleared to safe levels. This helps prevent cross-contamination between different medicines.
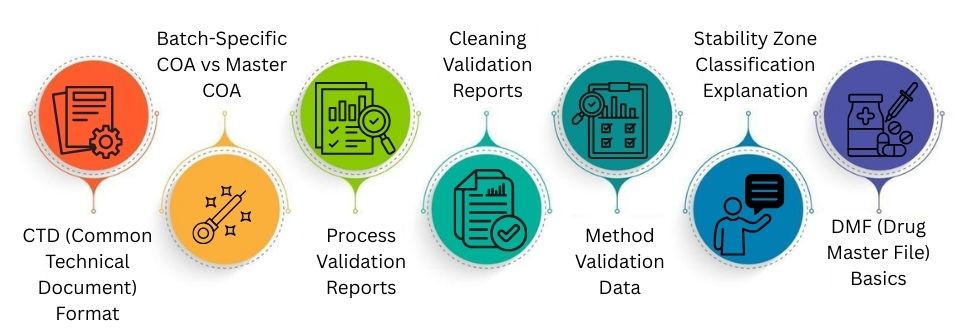
Method Validation Data
Method validation data shows that testing methods work well. They are accurate and reliable. It checks parameters like precision, accuracy, and detection limits. This ensures lab results are correct and trustworthy.
Stability Zone Classification Explanation
Stability studies show how medicines react to heat and humidity over time. Climate zones like I, II, III, and IVb guide storage conditions. Zone IVb covers hot and very humid countries.
DMF (Drug Master File) Basics
A DMF is a confidential file submitted to regulators. It covers details on manufacturing, raw materials, and controls. Buyers can reference it without seeing secret process information.
Change Control Procedures
Change control is a formal system for managing updates to processes, equipment, or methods. It makes sure that every change is reviewed, approved, and documented. This process maintains product safety and compliance.
A few groups review these documents before a purchase goes through. These include hospital groups, government organizations, and regulated distributors. Good documentation can make a big difference. It creates trust, speeds up approvals, reduces risks, and enhances your chances of landing major contracts.
Risk of Substandard & Falsified Medicines
Substandard and fake medicines are a serious global threat. Counterfeiting costs the industry about $200 billion every year. Below are the key risks and protection measures.
Substandard and fake medicines are a serious global threat. Counterfeiting costs the industry about $200 billion every year. Below are the key risks and protection measures.
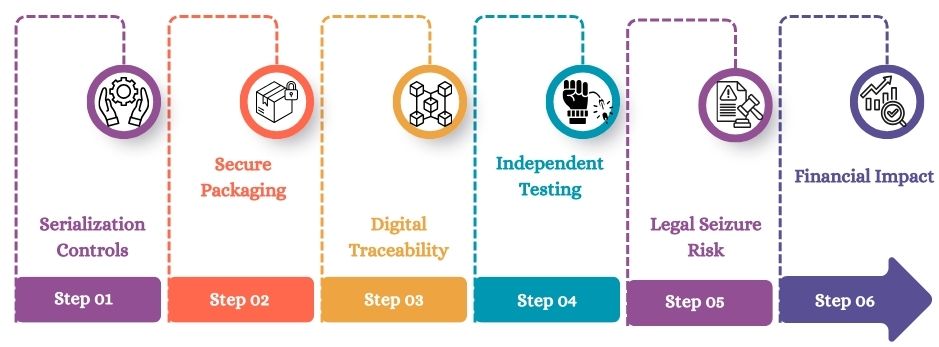
Serialization Controls
Serialization assigns a unique code to each medicine pack for easy identification. In India, QR codes became mandatory for APIs in January 2022. They are also required for the top 300 domestic brands since August 2023.
Secure Packaging
Tamper-proof packaging helps prevent product manipulation. Companies use security holograms, special films, and GS1 DataMatrix codes. These tools help protect packs and make copying or illegal refilling harder.
Digital Traceability
Track-and-trace systems record every step in the supply chain. Blockchain ledgers create a complete circle of trust. They record every ownership change, from the manufacturer to the final buyer.
Independent Testing
Third-party labs test products to ensure they are safe and meet quality standards. External verification cuts the risk of fake batches. This helps keep hospitals, pharmacies, and supply chains safe.
Legal Seizure Risk
Regulators can confiscate medicines suspected of being fake or substandard. These actions can stop shipments, delay tenders, start investigations, and may lead to penalties or a license suspension.
Financial Impact
Counterfeit exposure can lead to big financial losses and harm the brand. Companies risk losing contracts, and they may also face large fines. Their reputation with hospital groups and regulated distributors can suffer for a long time.
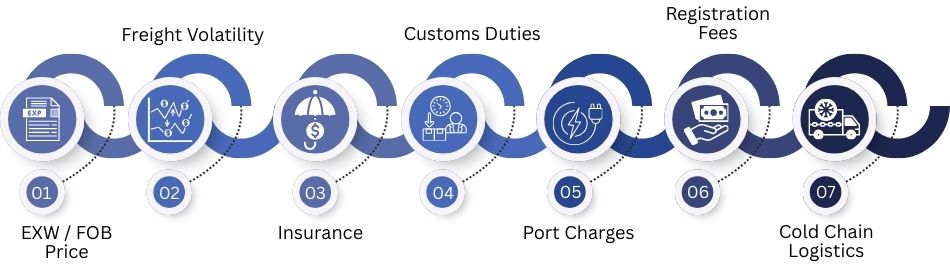
True Landed Cost Analysis: Beyond Ex-Factory Pricing
True landed cost is more than the factory price. Here are the key cost components, risks, delays, and new 2025 insurance and customs impacts.
Intellectual Property & Patent Risk Assessment
Intellectual property risk affects how companies decide to enter the generic market. It also impacts biosimilar market entries.
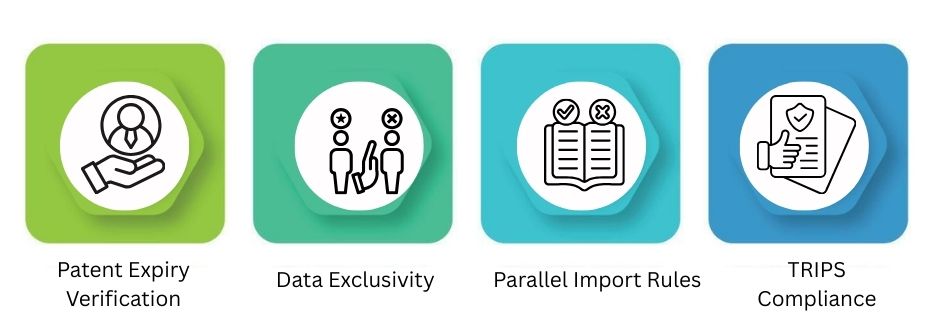
Patent Expiry Verification
To launch generic products, check the loss-of-exclusivity date. This includes secondary patents. It also covers extensions and ongoing court cases.
Data Exclusivity
Even after a patent expires, clinical trial data protection can still postpone approvals. This can block generics from hitting the market.
Parallel Import Rules
The rules governing the sale of patented medicines vary by country. National laws and international rules shape the resale policies in this context. These are often called “exhaustion policies” for short.
TRIPS Compliance
Countries generally meet minimum global IP standards. This gives some flexibility. It includes the Bolar provision and mandatory licensing for health emergencies.
Supply Chain Resilience & Multi-Sourcing Strategy
After COVID-19, companies changed how they buy and store goods. They now focus on safety and backup planning.

Here’s a brief overview of key concepts:
These are the suppliers:
This approach lowers dependency risk.
Country-Specific Import Risk Comparison
Import rules are not the same in every country. Some markets focus on inspections, others on safety reports or paperwork. Here’s a comparison of key regulatory risks in major countries.
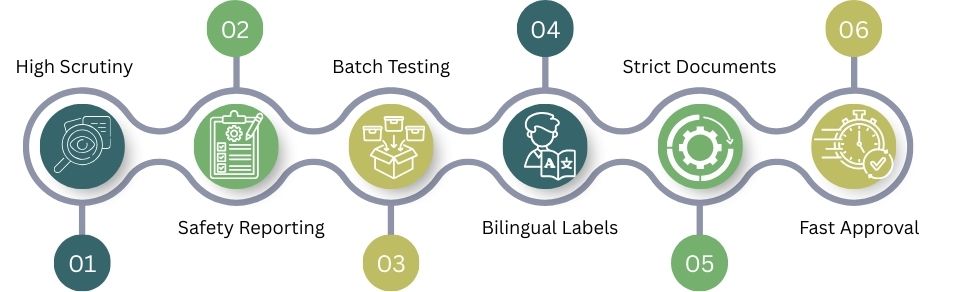
High Scrutiny
The U.S. Food and Drug Administration applies very strict inspections and GMP checks. Compared to most countries, the USA has a higher risk of rejection if compliance gaps are found.
Safety Reporting
The European Medicines Agency system and UK authorities stress strong pharmacovigilance. Unlike the USA’s inspection focus, they closely track side effects after products enter the market.
Batch Testing
Australia’s Therapeutic Goods Administration usually needs local batch testing. Unlike the UK and the EU, Australia places greater emphasis on pre-release lab checks for some medicines.
Bilingual Labels
Canada mandates English and French labeling. Compared to Australia or the USA, labeling errors alone can quickly stop shipments at the border.
Strict Documents
The UAE needs certificates that are officially recognized and verified. The UAE is strict about official documents. In contrast, Canada is more relaxed about paperwork.
Fast Approval
Singapore is known for efficient reviews. Manufacturers often receive approvals more quickly when they meet global quality standards. This is especially true compared to the US and Australia.
The spectrum starts with strict control over inspections and reporting. At the other end, you find efficient systems that follow the rules. The key difference is the country.
ESG & Sustainability Compliance (Advanced Section)
ESG compliance is now a core business need, not just a voluntary choice. Rules are becoming stricter each year. Below, we explain environmental standards, waste control, ethical sourcing, and future readiness.
Environmental Standards
Companies must follow strict environmental rules. They also need to provide audited sustainability reports. Clear data, third-party checks, and a way to measure impact are now essential. They help show responsible operations and ensure regulatory alignment.
Waste Control Systems
The main goal of modern waste management is to reduce waste. This is done by reusing and recycling materials whenever possible. Firms need to handle hazardous and chemical waste safely. They should track disposal digitally and aim to reduce landfill impact.
Ethical API Sourcing
To source APIs ethically, companies need to use green chemistry, audit their suppliers, and track everything. They also need to ensure that raw materials meet labor, safety, and environmental standards. This applies all along the supply chain.
Carbon Footprint
Carbon footprint awareness means knowing the emissions you create, both directly and indirectly. Businesses need to track supply chain emissions. They should set science-based targets and gradually reduce energy use.
Europe ESG Rules
The European Union is increasing its focus on ESG practices. It will enforce tougher disclosure rules and new steps to combat greenwashing. If you are exporting to Europe, you must provide verified sustainability data and clear reports.
Future-Ready Position
To stay competitive, suppliers need to show audit-ready ESG data. They should also have plans in place to cut carbon emissions and source materials responsibly. A good sustainability track record makes a supplier a trustworthy partner for the long haul.
Red Flags International Buyers Should Avoid
International buyers must check suppliers carefully before signing contracts. Small warning signs can lead to big losses later. The following explains common supplier risks and warning signals.
Suspicious Pricing
Prices that are much lower than usual may look like a bargain. But often, they signal the use of cheap materials, unknown outsourcing, or shortcuts that can compromise product safety.
No Audit History
If a supplier lacks an inspection history or avoids third-party audits, it’s a big warning sign for quality. This increases the risk of failing to meet compliance rules.
Missing Stability Proof
If a product’s stability data isn’t shared, it suggests the product may not have been fully tested. This includes how long it lasts on shelves, how it performs in different storage conditions, and its long-term safety in various climates.
Incomplete CPP
An incomplete or inconsistent Certificate of Pharmaceutical Product can hold up approvals. It might also raise worries about poor compliance or unclear documentation.
No Recall Plan
If a supplier doesn’t have a robust recall system, it’s tough to remove defective products quickly. That leaves buyers vulnerable to financial and legal headaches.
Unclear Logistics
When logistics aren’t clear, shipping routes can be confusing. If transport plans change suddenly, it becomes tough to track where products come from. This lack of clarity can lead to problems like delays, damage, or regulatory headaches.
Thorough due diligence is essential for buyers to avoid making costly mistakes. Check suppliers and review their documents. This helps you build trust and create strong, long-term partnerships.
Real-World Import Risk Scenarios (Case-Based Insights)
Importing goods involves legal, quality, and payment risks. Small mistakes can cause large losses. Below are explanations of real import problems and lessons learned from them.

Scenario 1:
A shipment was seized at the border due to patent violation in the destination country.
Lesson learned: always perform a freedom-to-operate check before exporting products internationally.
Scenario 2:
A pharmaceutical batch got rejected upon arrival. This happened because the stability data didn’t match the shipping conditions.
Lesson learned: check cold chain logistics and test products in the toughest transit conditions.
Scenario 3:
Goods were held at the port due to incorrect labelling and missing warnings.
Lesson learned: Always check label rules for each country. Do this before printing and sending shipments.
Scenario 4:
An importer paid the full advance to a new supplier and received delayed, poor-quality goods.
Lesson learned: Use safer payment tools, such as letters of credit, to reduce financial exposure.
Scenario 5:
A company failed to keep track of the temperature during sea transport. As a result, the products were damaged.
Lesson learned: Use continuous temperature data loggers. They help to ensure quality during long transit.
Scenario 6:
Missing or inconsistent regulatory documents caused delays in customs clearance.
Lesson learned: Always double-check compliance papers before shipping. This helps avoid storage charges and supply disruptions.
High-Growth Therapeutic Segments (2026 Outlook)
The 2026 outlook shows strong growth in key therapy areas. Below are the top segments and their expected growth rates based on IQVIA data.
In 2026, focusing on fast-growing areas will help with planning. It will also control costs and boost long-term success.
This is achieved through smart procurement strategies. There is a rising demand for lifestyle and chronic treatments. This has led to a rise in sourcing products worldwide from trusted suppliers like Fildena, Tadalista, Vidalista, Blue Zopiclone, and Generic Viagra.
Structured Due Diligence Framework That Reduces Import Risk by 60%+
Importing medicines or APIs has legal, quality, and financial risks. Here’s a simple eight-step framework to reduce risks. It boosts compliance and improves supply security.
A system set up in a structured way really pays off because each step assesses a different potential risk. By following the steps in order, you can avoid compliance gaps, minimize surprises, and build solid, lasting relationships with your suppliers.
Why India Remains a Strategic Long-Term Partner
India continues to grow as a trusted global supplier. Here are the six key strengths that support long-term partnership stability and growth:
Large-Scale Production
India operates numerous manufacturing units in the pharmaceutical and chemical sectors. High production capacity means more output. This leads to faster turnaround times and lets us serve many buyers, from small to large, around the world.
Skilled Talent Base
The country has a young, trained, and science-focused workforce. Engineers, chemists, and technical experts support manufacturing. They also assist with quality control and research. All activities follow international industry standards.
Lower Production Costs
Manufacturing costs in India are 30–70% less than in many Western nations. Lower labor, infrastructure, and operations costs lead to higher margins for buyers. This happens without sacrificing product quality.
Global Quality Standards
Indian companies follow strict international rules. They also align with major global authorities. This regulatory alignment cuts approval delays. It also boosts confidence for exports to regulated markets.
Robust API Network
India has a solid system for Active Pharmaceutical Ingredients. It is well-organized and efficient. The country’s industrial clusters are a key asset. They offer key raw materials, skilled workers, and dependable sourcing support.
Export Policy Support
Government programs help manufacturing and exports. They do this by giving financial incentives and also supporting infrastructure. These initiatives boost global competitiveness. They also help companies grow sustainably in global markets.
Strategic Conclusion: Importing Medicines Is a Risk-Managed Partnership
Importing medicines is more than just buying products. Here are the five pillars of safe and stable sourcing partnerships.
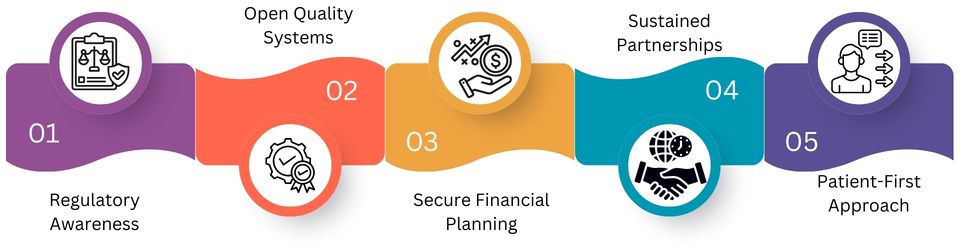
Regulatory Awareness
Solid compliance systems keep up with changing global rules and standards. This cuts down approval delays and avoids penalties. It also ensures that imported medicines comply with safety and quality guidelines.
Open Quality Systems
Transparency is key when it comes to building trust. Buyers want easy access to manufacturing data, batch reports, and testing results. This helps them confirm that products meet standards and are safe.
Secure Financial Planning
Thorough supplier vetting, clear contracts, and secure payment terms can reduce financial risks. It’s about being prepared to minimize potential losses. This safeguard protects your working capital. It also helps you maintain a stable and cost-effective supply chain with fewer issues.
Sustained Partnerships
Long-term relationships improve communication, planning, and reliability. Strategic collaboration reduces market shocks, shipping delays, and supply gaps. It also boosts overall procurement stability.
Patient-First Approach
Patient health is the top priority in sourcing decisions. We need robust tracking systems and regular safety checks to ensure medicines remain safe and effective. We also need to keep a close eye on their quality to ensure they’re reliable.
Importing medicines works best when there is a clear partnership and simple processes. With the right systems, buyers can protect patients.
They can also safeguard their investments and reputation. This helps create a strong and secure supply model for the future.
