Sorry, nothing in cart.
The global pharmaceutical supply chain is key for buyers. It shows how medicines move from factories to patients. India supplies about 20% of global generics and exported over $25.3 billion in FY2023.
Strong manufacturing and reliable suppliers ensure safe global distribution. Quality checks help maintain steady access to affordable medicines.
Pharmaceutical suppliers manage sourcing, compliance, and cross-border shipping. The supply chain management market is expanding. It will rise from around $2.85 billion in 2025 to $4.22 billion by 2030.
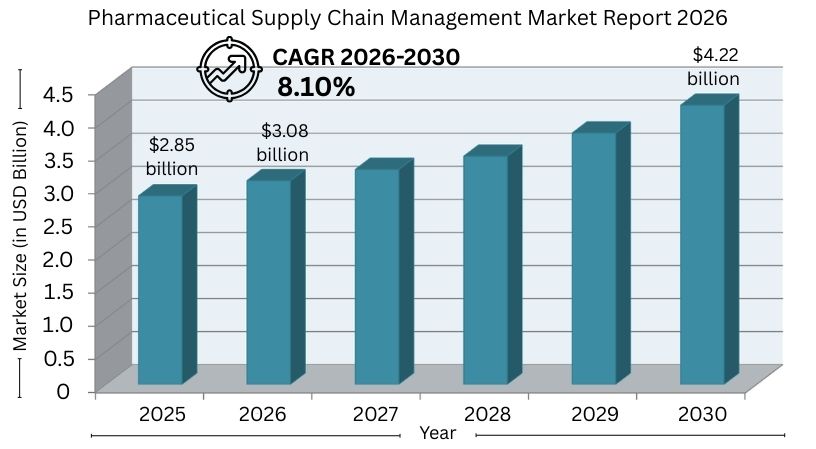
This growth comes with an 8.10% CAGR. Current trends are multi-sourcing, digital tracking, and cost-efficient procurement. These can save 30–35% by using large-scale production regions.
Evaluate Supplier Credibility & Reputation
Choose your suppliers wisely. This keeps medicines safe and offers easy shipping. It also builds reliable partnerships worldwide, especially with trusted partners like Fildena Wholesaler, Cenforce Supplier, and Vidalista 20 Supplier.
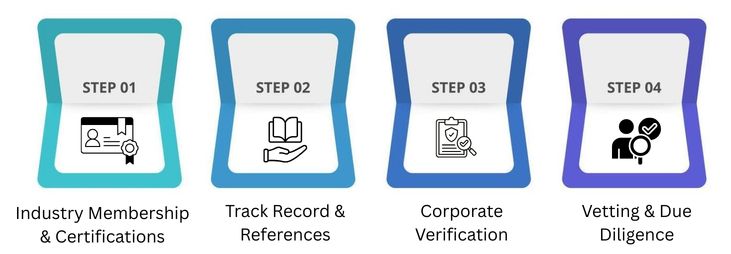
Industry Membership & Certifications
Check if the supplier is in the Pharmaceuticals Export Promotion Council of India (Pharmexcil). Look for other similar trade bodies too.
Membership shows export compliance and access to official data. Check GMP approvals and quality certificates before you place orders.
Track Record & References
Review years in business, client feedback, and global shipment history. Reliable exporters often work with NGOs or UN programs.
Ask for references. Request past project reports. Also, seek proof of regular international deliveries.
Corporate Verification
Check company details using records from the Ministry of Corporate Affairs. Check GST, PAN, IEC numbers, and director information. Make sure business addresses and legal status are valid and up to date.
Vetting & Due Diligence
Work with suppliers who audit their manufacturers. These suppliers need to source only from GMP-certified plants. Request CoA, GMP certificates, and batch documents. Good vetting stops bad goods. It also prevents shipping problems and customs denials.
Regulatory Compliance & Certifications
Strict regulations help buyers shop safely and maintain high quality. They also help trusted pharmaceutical companies, including Vidalista Supplier and Tadalista 60 Wholesaler, sell their products worldwide.
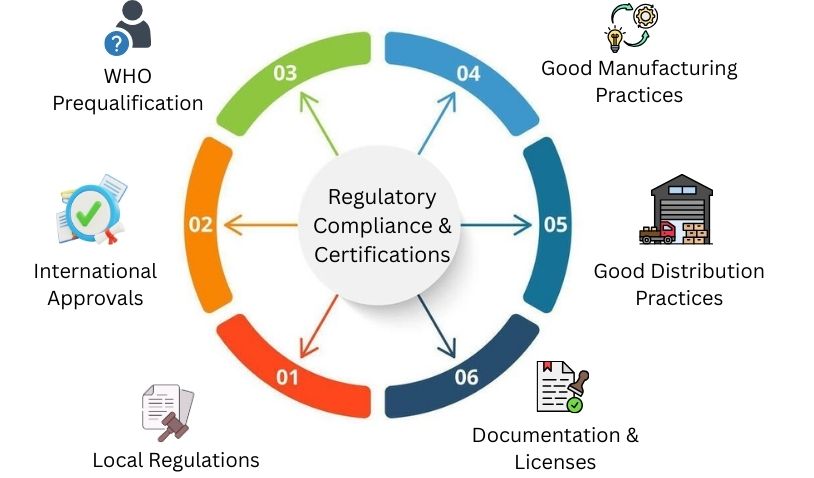
Local Regulations
Suppliers must follow the rules. These rules are issued by India’s Central Drugs Standard Control Organization.
They must follow the Drugs and Cosmetics Act. They need valid licenses and export permits for legal manufacturing and trade.
International Approvals
Check for certifications from these agencies:
This shows that the global market accepts and complies with inspections.
WHO Prequalification
WHO standards check quality. Prequalification makes sure systems are up to standard. India reports 2,050 WHO-GMP plants. It also has hundreds of facilities approved worldwide.
Good Manufacturing Practices
cGMP ensures safety in production. It uses modern systems and quality checks. India has 752 FDA-registered and 286 EDQM-approved sites (2024).
Good Distribution Practices
GDP guidelines keep products safe while they are stored and transported. WHO TRS 1025-based drafts guide India’s supply chain partners and logistics teams.
Documentation & Licenses
Filling out paperwork, like COAs, licenses, and export records, helps avoid delays. Wrong or missing documents can cause shipment rejections. This can happen anywhere in the world.
Quality Assurance & Pre-shipment Checks
Quality checks make sure buyers get safe medicines. They also ensure that the medicines comply with regulations.
They also help avoid shipment delays, recalls, and regulatory issues in different markets.
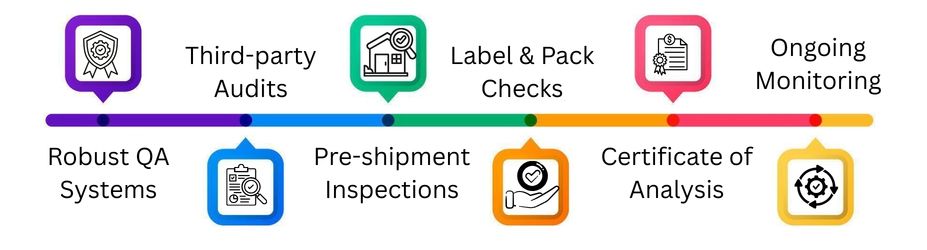
Robust QA Systems
Suppliers must follow strict GMP/PIC/S standards. They should validate their processes, check everything as they go, and test each batch in a proper lab.
Third-party Audits
Independent or FDA-style inspections look at manufacturing sites. They check for real production quality. They also check for compliance before making sourcing decisions.
Pre-shipment Inspections
Products should be checked before dispatch. Industry data shows inspections can reduce non-compliance risks by over 45% during export.
Label & Pack Checks
Pre-dispatch reviews check labeling, packaging, and specifications. They make sure these items meet regulatory rules. This helps avoid shipment rejections and costly product recalls.
Certificate of Analysis
Each batch needs to have a signed Certificate of Analysis. This shows it meets specs, checked by trained staff who know what they’re doing.
Ongoing Monitoring
Suppliers should track batch integrity using serial numbers or barcodes. They can use digital systems to avoid mix-ups, counterfeits, and supply errors.
International Certifications & Accreditations
Global certifications build trust. Suppliers like Cenforce Supplier, Fildena Wholesaler, and Kamagra Oral Jelly Wholesaler comply with WHO, USFDA, EMA, ISO, and PIC/S standards.
Look for:
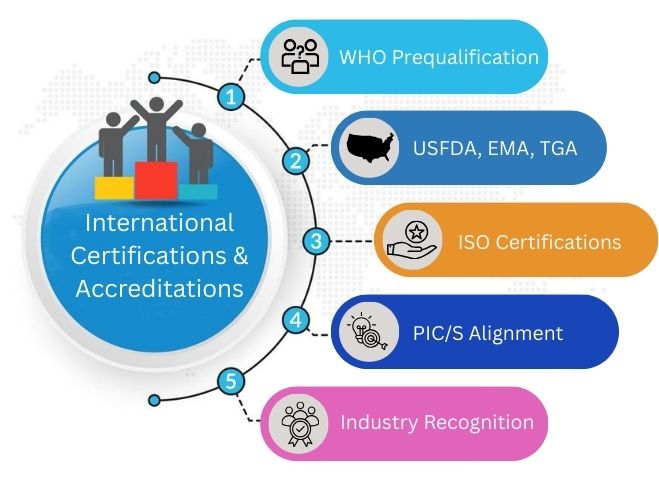
WHO Prequalification
The World Health Organization checks quality. India has around 2,050 WHO-GMP-certified plants that support global procurement.
USFDA, EMA, TGA
The U.S. Food and Drug Administration, European Medicines Agency, and Therapeutic Goods Administration give approvals. These approvals allow entry into strict global markets.
ISO Certifications
ISO standards, such as ISO 9001, support quality systems. They also ensure consistent manufacturing across facilities.
PIC/S Alignment
Countries in the Pharmaceutical Inspection Co-operation Scheme have the same GMP rules. Each country follows these rules closely.
This ensures consistency in quality. This makes inspection quality better. It also builds global trust in regulations.
Industry Recognition
Awards highlight quality leadership. As of 2024, India has 752 sites registered with the FDA. It also has 286 facilities approved by EDQM. This supports its global credibility.
Exported Product Portfolio
Production Capacity & Supply Reliability
Strong production capacity and a reliable supply help companies avoid delays. They can manage changes in demand. This helps to keep operations running smoothly.
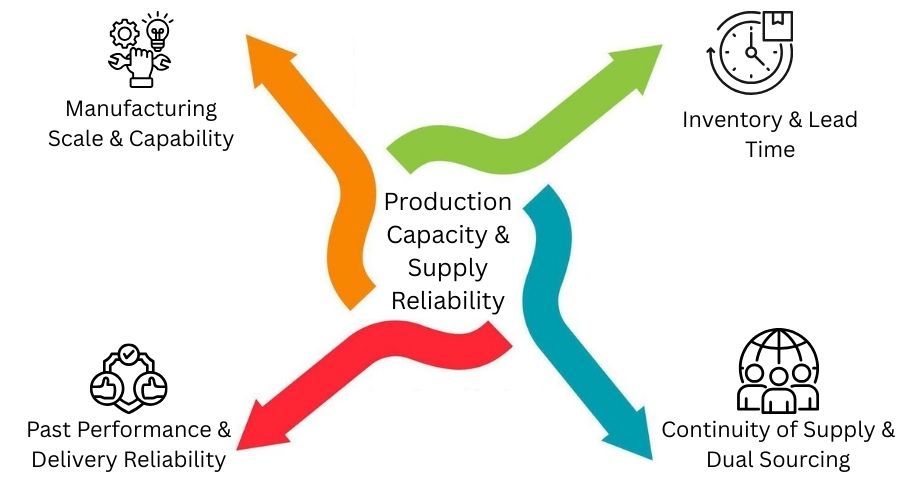
Manufacturing Scale & Capability
A good manufacturer should manage large and small batches.
They must also ensure quality stays steady.
Modern gear, trained teams, and flexible operations help meet sudden demand spikes. At the same time, they keep things consistent and compliant.
Inventory & Lead Time
Clear inventory planning and safe stock levels reduce shortages. Short and predictable lead times help buyers plan well. This keeps production steady and avoids emergency orders or delays.
Continuity of Supply & Dual Sourcing
Using primary and backup suppliers lowers risk. Using dual sourcing from different areas helps keep the supply steady.
It also helps during disruptions, including problems such as logistics issues. It also includes shortages of raw materials and sudden shifts in demand.
Past Performance & Delivery Reliability
Check past performance by looking at delivery records, shipping times, and quality outcomes.
A supplier is reliable if they often meet expectations and reply quickly. These traits prove they can be trusted for long-term supply stability.
Logistics Infrastructure
Strong logistics infrastructure keeps products safe. It protects quality and cuts delays. This ensures products move easily from warehouses to customers.
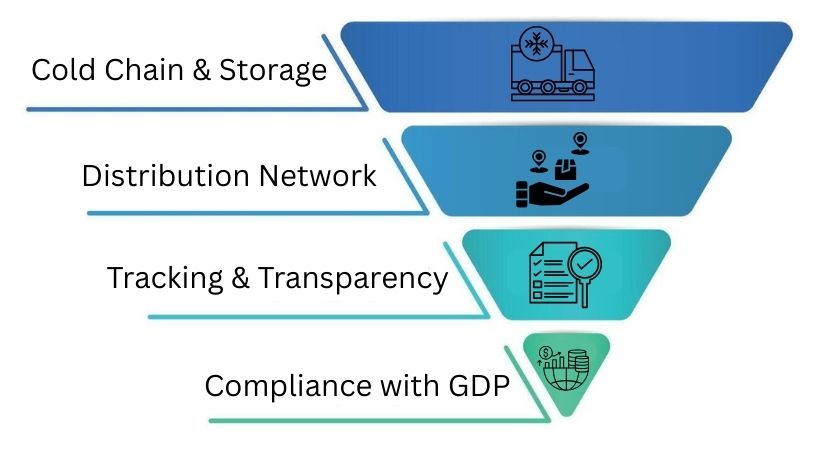
Cold Chain & Storage
Temperature-controlled storage keeps sensitive products safe. This applies during transit and storage. To keep things at the right temperature, proper packaging and careful cooling checks are a must.
Trained teams are also on the job to prevent damage to the products.
Distribution Network
A solid delivery network is crucial for getting items to customers quickly.
It performs well across various regions. This setup helps reduce the risks that come with shipping. Set routes also improve supply coverage.
Tracking & Transparency
Real-time tracking tools show clear updates.
They track shipments from dispatch to delivery. Digital records, alerts, and shared dashboards let buyers track orders. This way, they can respond quickly to any delays.
Compliance with GDP
Good Distribution Practices help keep handling safe. They also ensure safe storage and transport. Good documentation and trained staff are important.
Regular audits help ensure compliance. They also protect product quality in the supply chain.
Cost-Effectiveness & Value
Cost planning should prioritize long-term value over low prices. This method keeps supply partnerships steady and efficient.

Price vs Quality Balance
Choosing the lowest price may risk quality and delays. A balanced supplier provides fair prices and steady quality. This helps prevent rework, returns, and production delays.
Total Landed Cost
True cost includes the product price. It also covers freight, insurance, customs duties, and handling fees.
Calculating the total landed cost helps you compare suppliers better. It also stops unexpected expenses after you place an order.
Flexible Pricing & Payment Terms
Flexible pricing models and payment terms support better cash flow.
Options like bulk discounts, staged payments, and credit terms help buyers manage their budgets. They also build long-term trust with suppliers.
Communication & Transparency
Clear communication prevents confusion. It builds trust and keeps supply partnerships smooth and reliable.
The information should be shared so that work runs smoothly and problems are avoided in the following way:
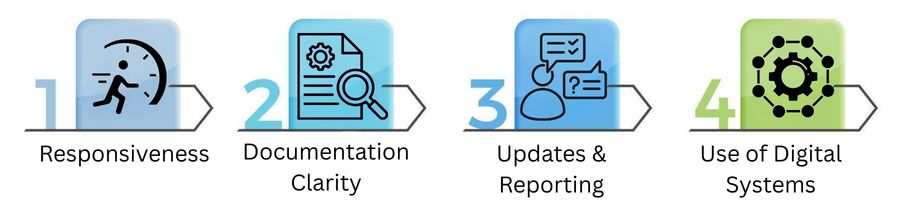
Responsiveness
Fast replies to emails, calls, and queries help solve issues early. A responsive supplier updates quickly and answers clearly. They also support buyers during urgent production or shipping needs.
Documentation Clarity
Clear documents reduce mistakes and delays. Clear invoices, certificates, and shipping papers help teams. They make it easy to understand orders.
This reduces confusion during audits or inspections.
Updates & Reporting
Regular updates on production, stock, and shipments keep buyers in the loop. Simple progress reports and alerts help teams plan well. They also let teams respond quickly to any delays.
Use of Digital Systems
Digital tools such as tracking portals, shared dashboards, and automated alerts boost visibility. These systems help teams get real-time data.
They also make order management more accurate.
Risk Mitigation Strategies
Good risk planning protects supply chains. It cuts down disruptions and keeps production stable during surprises or changes.
Dual Sourcing
Using two qualified suppliers reduces dependence on one source. Adding safety stock buffers helps manage delays. They also help with demand spikes and transport problems. This way, production can keep running smoothly.
Contract Safeguards
Clear contracts set quality standards, delivery timelines, and responsibilities. Service levels, penalties, and backup plans help buyers. They protect against delays and supply problems.
Regular Audits
Regular checks on suppliers help ensure they meet standards.
This includes quality, capacity, and compliance. Reviewing performance data and site visits helps spot risks early.
This way, we can prevent issues and ensure suppliers are more reliable over time.
Regulatory Watch
Staying on top of regulatory updates, trade rules, and safety standards really pays off. It helps to avoid surprises and keeps your supply chain working well.
Operational Maturity & Technical Capacity
Operational maturity shows how well a supplier manages quality.
It measures their ability to maintain standards. It also reflects their handling of staff and technology. This management helps ensure consistent, safe production.
Facility Hygiene & Environmental Control
Clean and controlled spaces are key to reducing the risk of contamination.
It’s all about maintaining good air quality, cleaning regularly, and keeping a close eye on things. This keeps product quality safe during production and storage.
Equipment Validation & Calibration
Using the right gear is key to accurate, repeatable results. Regular checks are essential to achieve this.
Preventive maintenance helps prevent issues before they occur.
Also, remember to keep a record of your tests. This all helps ensure that things work reliably and reduces the risk of unexpected breakdowns.
Personnel Competency & Culture
Trained teams organize procedures. They also focus on quality practices. This helps staff catch issues early on and stick to consistent manufacturing standards.
Ongoing learning, a strong safety culture, and accountability for actions are all key to making this work.
Technology in Supply Chain
New tech is giving our tracking capabilities a major boost. Advances like serialization and digital tracking are changing things.
AI analytics and blockchain are also making a big difference. They help people see things clearly, avoid mistakes, and decide faster in the supply chain.
Verification & Legitimacy Checks
Proper verification makes sure suppliers are real. It also checks if they follow rules and are stable. This cuts risks before long-term partnerships start.
Key supplier verification and compliance can be done using:
Databases & Portals
Check trusted sites like EudraGMDP, WHO Prequalification, and U.S. FDA records. These databases confirm approvals and inspections. They also track registrations for manufacturers and products worldwide.
QMS & Data Integrity
A good pharmaceutical quality system is key to keeping track of records and making data easy to follow.
With secure access controls, audit trails, and clear procedures in place, you can safeguard accuracy and prevent unauthorized data changes.
Financial Due Diligence
Review a supplier’s finances, production skills, and history. If they’re stable, have a solid cash flow, and a long history, they’re probably more reliable when the market gets bumpy.
Ethics & IP Protection
To keep partnerships safe, companies need clear rules. They should focus on preventing bribery and misuse of information.
Contracts should include confidentiality agreements and IP protection. They also need clear rules for fair conduct. This helps maintain trust and ensures everyone follows the rules.
Global Logistics & Customs Compliance
Strong logistics and customs planning are vital. They help keep product quality high. This reduces wait times and helps international shipments run more smoothly.
Temperature-Controlled Infrastructure & Monitoring
Good cold chain systems keep products at safe temperatures. They ensure items stay cool and fresh. They do this during storage and while moving.
To stop temperature changes and quality issues, companies use tools. These tools watch conditions all the time. They rely on trusted packaging and logistics partners who really know their stuff.
International Trade & Customs Clearance
Filling out export documents is important. It helps your shipment clear customs quickly. Following trade rules also speeds up the process.
Work with experienced partners who can handle duties, permits, and inspections for you. This can save time and make cross-border delivery a whole lot smoother.
Supplier Evaluation Process
Evaluating suppliers carefully helps you choose partners you can count on. This process also manages risks and keeps quality and delivery performance steady.
Scorecards & KPIs
Make easy supplier scorecards using key performance indicators. Focus on quality, how quickly they deliver, how they respond to issues, and cost control.
Regular reviews track performance. They also support better decisions.
Virtual & On-Site Audits
Start with virtual audits to review documents and systems. Check facilities, processes, and teams with on-site visits. This ensures that real operations match the information shared.
RFP Best Practices
Create clear and simple RFPs that outline specific needs and scoring methods. Using standard templates helps keep things fair. Clear criteria make it easier to compare suppliers. These tools also help you pick the best partner.
Sample & Pilot Testing
Before giving full approval, test out a few product samples and run a small pilot order. This makes sure the quality is good, the packaging is strong, and deliveries run smoothly.
Negotiation & Contracts
When negotiating, watch for the price, service levels, and delivery terms. A good contract should clearly state responsibilities.
To maintain strong relationships with suppliers, clearly state your quality expectations. Then, outline the consequences for not meeting these standards.
Also, explain how you will handle confidentiality.
Tools & Technologies
Modern tools boost visibility, safety, and planning in supply chains. They help teams manage risks and keep operations running smoothly.
Serialization & Track-and-Trace
Special codes on each product let you track them from the factory to the customer’s doorstep.
These track-and-trace systems provide a clear view of location. They stop fake products from being sold and help you meet global supply chain rules.
Blockchain & IoT Integration
Connected sensors and digital ledgers record things like location and temperature.
They also track how products are handled. This info builds trust and keeps data safe. It gives you a clear record of how products were moved and stored.
AI & Predictive Analytics
AI tools study data to predict delays, shortages, or risks. Predictive analytics help teams plan ahead, adjust orders, and respond faster to changes across global supply networks.
Long-Term Supplier Management
Building long-term relationships with suppliers pays off. You ensure high quality, lower risks, and keep the supply chain stable. Regular check-ins and updates make this possible.
Ongoing Monitoring
Continuous monitoring is key to making sure delivery, quality, and service stay on track. Regular audits and review meetings help spot issues early. This allows suppliers to improve their performance.
Regulatory Updates
Regulatory rules and standards are always changing. To keep suppliers updated, check in with them regularly.
Also, make sure documents are current and share any compliance alerts. This helps make sure they meet the latest requirements and certifications.
Scorecard Reviews
Using supplier scorecards helps you check quality. They also track delivery time and responsiveness. Regular check-ins of these scorecards help you make fair calls and push for better performance over time.
Conclusion
To pick a trusted pharmaceutical supplier, you need a smart strategy. This ensures safe and reliable global distribution.
Buyers should mix strong compliance checks with quality assurance systems. They also need steady supply planning and ethical practices.
This mix helps everything run smoothly. Users should carefully plan and perform checks to ensure quality.
A strong partnership relies on a few simple things: clear communication, the latest tech, and regular check-ins.
When compliance, quality, and ethics are in sync, supply chains run smoothly. This makes them reliable. This helps them deliver medicines safely in global markets.
Reach Out on Social Media
Stay connected and get the latest updates from our pharmaceutical suppliers and global supply chain insights.






