Sorry, nothing in cart.
What Is Triclabendazole and Why Does It Matter?
TCBZ, or Triclabendazole, is an anthelmintic. It is made from benzimidazole. The World Health Organization recommends it for the treatment of human fascioliasis. It helps eliminate parasitic worms, which is why they suggest using it, and Triclabendazole 250 mg (Triclabend) is one of the commonly used strengths for this treatment.
Liver flukes, specifically Fasciola, cause this disease. This disease is often neglected and found in tropical areas.
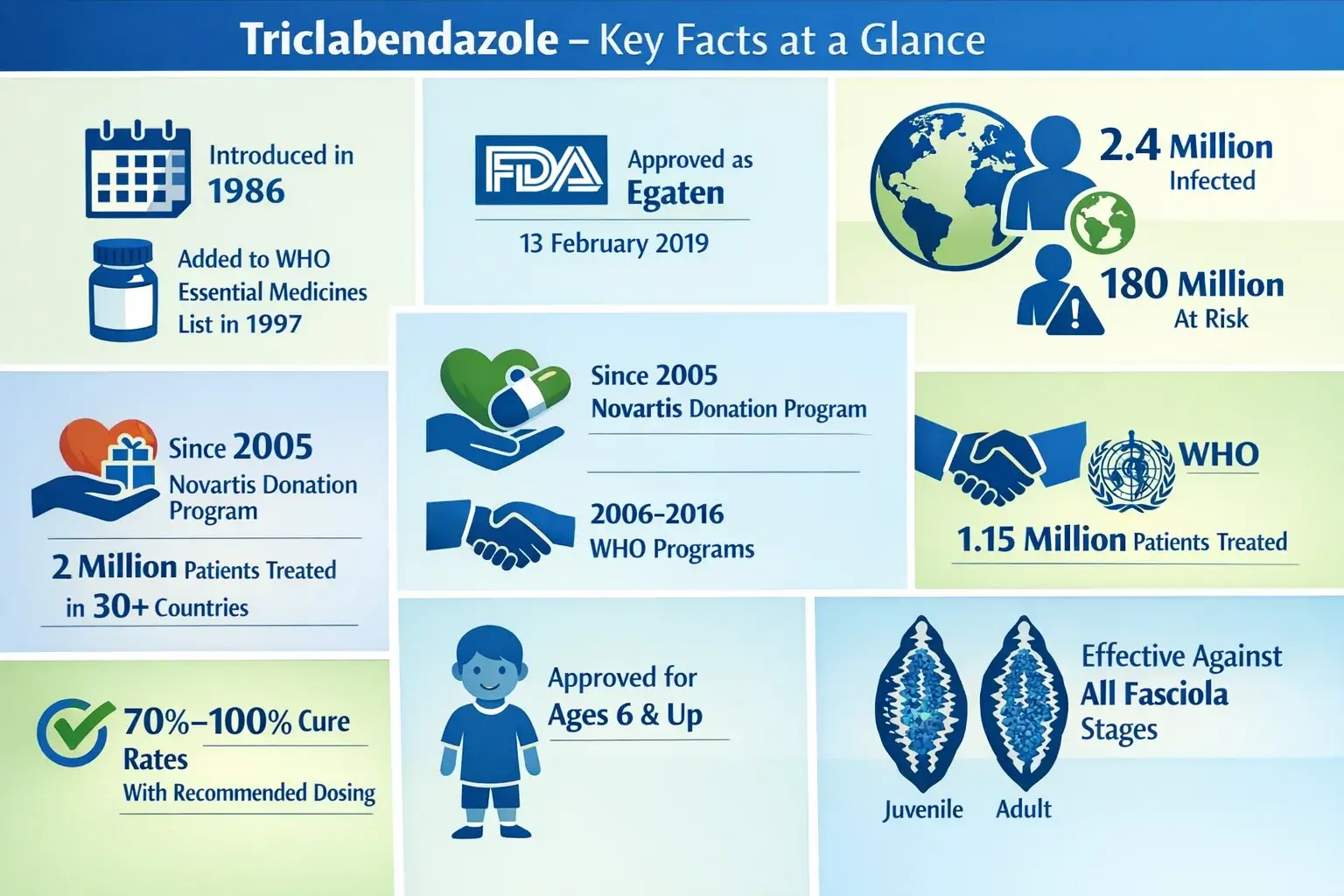
It’s been the go-to drug since 1986, thanks to its high success rate in treating patients. Even though some areas are concerned about resistance, it remains the top choice.

Fascioliasis affects around 2.4 million people globally. On top of that, approximately 180 million people are at risk of contracting it on every inhabited continent.
It’s common in countries such as Peru, Bolivia, Egypt, Iran, and Vietnam. In kids, this can trigger fever, anaemia, and developmental problems. If infections are not treated, they can cause serious liver problems. So, getting the right treatment is crucial.
Triclabendazole – Key Facts at a Glance
Triclabendazole is really important for global health. There’s a lot of treatment data, along with major regulatory milestones, that support this. It’s a key player in the field, and that’s clear from the numbers and the milestones it’s hit.
Mechanism of Action: How Triclabendazole Works
Triclabendazole treats liver flukes from the inside out. It targets the infection directly, stopping it at its source.
This medication treats fascioliasis well. It works whether the condition is caught early or late. This helps doctors and makes it easy for patients to understand simple treatment explanations.
The drug harms important parts of the parasite. This leads to its eventual death. It stops movement, feeding, and reproduction. This keeps the infection from spreading. It works on many stages of parasites. This makes it one of the most reliable treatments today.
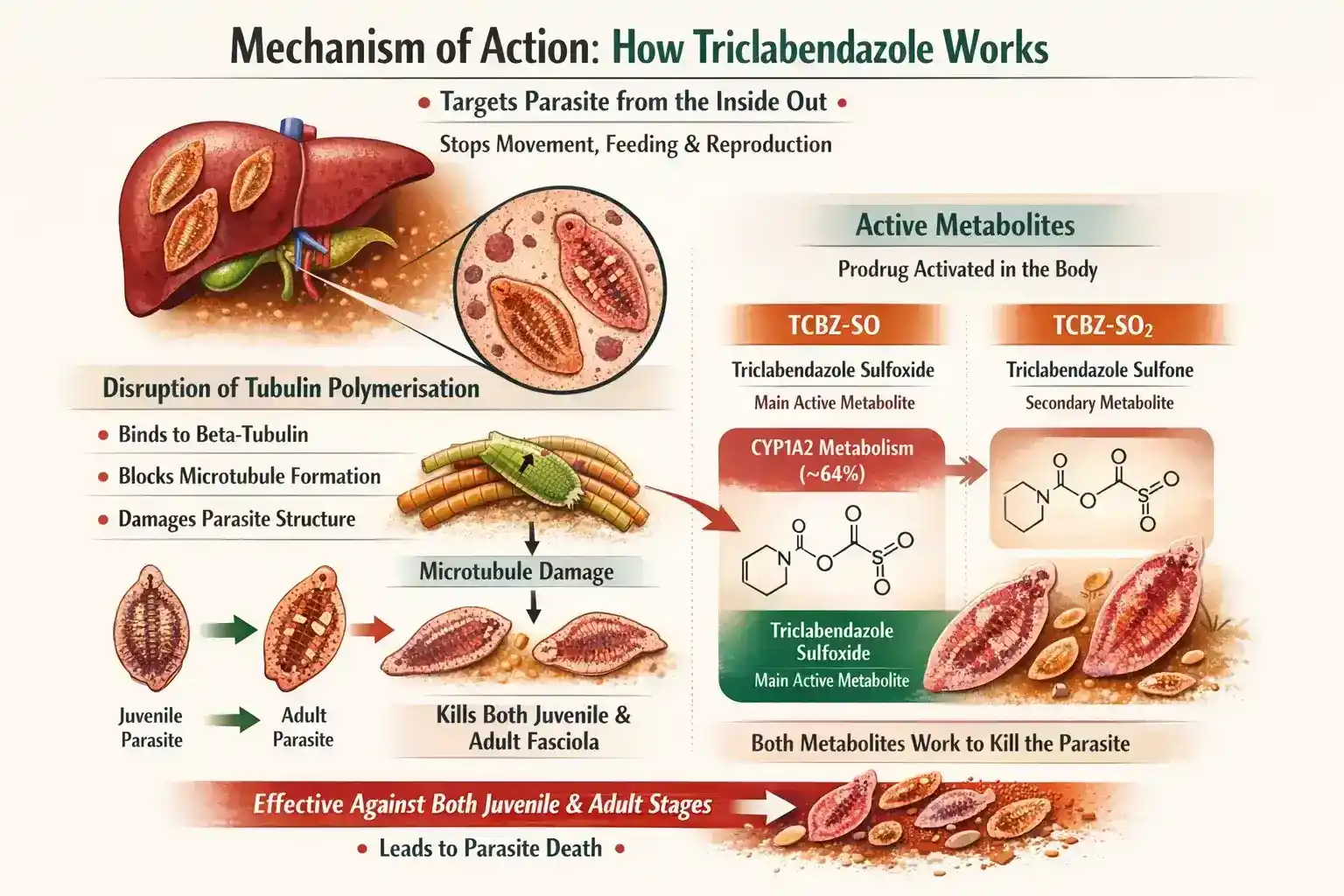
Disruption of Tubulin Polymerisation
Triclabendazole attaches to beta-tubulin in the parasite.
This prevents the creation of microtubules. This damages the parasite’s structure and disrupts key functions. These include moving, absorbing nutrients, and producing eggs. As a result, it can’t survive.
A big plus is that it can kill both juvenile and adult Fasciola. This is rare for anthelmintic drugs.
Active Metabolites: Sulfoxide (TCBZ-SO) and Sulfone (TCBZ-SO2)
Triclabendazole is a prodrug, so it doesn’t work until it’s inside the body. It becomes active once it’s absorbed.
It is mainly metabolised by CYP1A2 (around 64%) into sulfoxide (TCBZ-SO).
This is the main active form, with the highest blood levels after dosing. This is converted to the sulfone (TCBZ-SO2). Both metabolites work together to kill the parasite effectively.
Pharmacokinetics Table (FDA Label Data)
| PK Parameter | Key Values |
|---|---|
| Cmax Sulfoxide (TCBZ-SO) | 38.6 μmol/L |
| AUC Sulfoxide | 386 μmol·h/L |
| Primary Metabolism | CYP1A2 ~64% |
| Excretion | Biliary/feces ~90% |
| Food Effect | ↑ Bioavailability 2-3× – MUST be taken with food |
To get the most out of Triclabendazole, it’s best to take it with food. This way, your body can absorb it more easily.
Critical Clinical Note: Eating food with it makes a big difference. It is about 2-3 times more effective. If you don’t take it with enough food, it might not work well. This can cause the treatment to fail.
Where to Buy Triclabendazole for Humans?
When it comes to treating fascioliasis, the best and first-line treatment suggested is Triclabendazole Tablet. The medication helps to overcome infection easily by protecting your overall health. But the concern among individuals is – where to buy Triclabendazole?
Now therein, the role of a trusted supplier comes, and one big name in the pharma industry is Actiza Pharmacy.
At Actiza Pharmacy, we offer Triclabendazole Tablet and Triclabendazole 250mg for individuals seeking safe and effective treatment options at much affordable rates. Our supply is fast and reaches across different countries, adhering to all necessary guidelines, compliance, and regulations.
Triclabendazole has been a safe and reliable tablet for the treatment of fascioliasis among humans. Available in 250 mg strength, the dosage is given depending upon the individual’s body weight, severity of infection, and other health conditions. Triclabendazole works faster to eliminate the liver fluke and must be consumed under the complete supervision of a specialist.
Recommended products
Regulatory History and FDA Approval
This section shows EEAT credibility. It highlights Triclabendazole’s 25-year journey from synthesis to FDA approval.
It underwent lab research, safety testing, and human trials. This process ensured the medicine was effective and safe for long-term use.
Key Regulatory Milestones Timeline Table
| Year | Milestone |
|---|---|
| 1978 | Synthesised by Ciba-Geigy (now Novartis) |
| 1983 | Veterinary use as Fasinex (livestock) |
| 1986 | First human use (Europe) |
| 1989-92 | WHO clinical trials – 6 trials, 5 countries: Bolivia, Chile, Peru, Cuba, Iran |
| 1997 | Approved Egypt (human use); Added to WHO Essential Medicines List |
| 2002 | EU approval – France (first EU country) |
| 2005 | Novartis begins WHO donation program for endemic countries |
| 2006-2016 | 1.15 million patients treated through WHO donation |
| Feb 13, 2019 | US FDA approved EGATEN – first FDA approval for fascioliasis |
| 2019 | Orphan Drug Designation + Fast Track granted by FDA |
The FDA review checked out data from over 900 patients. It covered a lot of ground, including published studies, compassionate use, and some named programs. It confirmed that Triclabendazole is safe and has no major concerns. This boosts global confidence in the drug.
Approved Dosing and Administration
This section shows how to dose Triclabendazole. It follows FDA guidance. It also makes it easy to compare with the practices of the WHO and the CDC.
It explains how to take the drug, including when to eat, for safe use and better treatment results for different patient groups.
Official Dosing: Egaten (Triclabendazole) – FDA Label
WHO vs CDC vs FDA Dosing Comparison Table
Health authorities suggest various dosing plans. These depend on the setting and public health needs. The goal is always clear: to remove parasites safely and effectively. They want to make sure the treatment works and doesn’t harm anyone.
In simple terms, WHO might pick single-dose strategies. They could use these for large programs. The CDC and FDA recommend giving two doses for better treatment outcomes. This helps achieve better cure rates and lowers the chance of incomplete treatment.
Clinical Data: Cure Rates and Treatment Outcomes
This section outlines the key clinical findings on Triclabendazole. It focuses on cure rates and treatment outcomes, giving you an idea of its effectiveness.
It helps readers understand how effective the drug is across studies and in real patient use.
Pivotal Clinical Trials Table (1989-2019, 11 studies)
| Study / Country (n) | Regimen → Cure Rate |
|---|---|
| Bolivia – 2 trials (~40 patients) | 10 mg/kg single/double → 70-100% |
| Chile (~20 patients) | 10 mg/kg → 85-100% |
| Peru (~20 patients) | 10 mg/kg → 80-100% |
| Cuba (~20 patients) | 10 mg/kg → 70-100% |
| Iran – WHO 1989-92 (~107 patients) | 1, 2 or 3× 10mg/kg → 63.9-68.6% |
| Vietnam RCT (100 patients) | 2× 10 mg/kg → 92% symptom-free at 3 months |
| Egypt – El-Morshedy (134 patients) | 1 dose: 79.4% cure | 2 doses: 93.9% cure |
| Peru RCT – Paediatric (84 children) | 2× 10 mg/kg → High efficacy |
| Bolivia Community (90 children | 2× 10 mg/kg → High efficacy |
| Iran – Chronic (165 patients) | Multiple regimens → Dose-dependent cure |
| OVERALL – 2-dose regimen | 20 mg/kg total → 95.5% cure at Day 60 (FDA basis) |
Triclabendazole proves to be highly effective. The cure rates remain strong and consistent across many studies. The two-dose regimen gives the best results.
This supports its status as the standard treatment for fascioliasis worldwide.
Global Treatment Outcomes by Country / Region
This section shows country-wise treatment outcomes, most important for SEO and international patients, covering seven countries with real-world cure rates and effectiveness across different regions.
Peru (Andes) – Highest Global Burden
Peru’s Andean regions have a high rate of fascioliasis. This is especially true for children. This area is one of the worst-affected in the world.

Bolivia – World’s Largest National Program
Bolivia has the biggest triclabendazole program. It aims to treat fascioliasis.
This program helps high-risk Andean communities. Below, key program details explain the large-scale treatment strategy, target groups, and global public health impact.

Egypt (Nile Delta) – First Large-Scale Public Health Intervention
Egypt launched a major effort to control fascioliasis. They used Triclabendazole in high-risk parts of the Nile Delta.

Here are key milestones:
Iran (Caspian Lowlands) – Major Outbreak History
Iran has a complex history with fascioliasis. There have been major outbreaks in the Caspian lowlands that have hit thousands of people.

Below are the key data highlights:
Vietnam – Only RCT in FDA Evidence Base
Vietnam ran the only randomised trial in the FDA data. It showed that Triclabendazole works. Below, key trial results compare treatments and show clinical outcomes.

Only RCTs are included in the FDA’s evidence base for EGATEN approvals.The study included 100 patients. They all had acute symptomatic fascioliasis.
Triclabendazole had a 92% symptom-free rate after three months.Artesunate had a 76% symptom-free rate, which is lower than that of Triclabendazole.
Results showed that Triclabendazole worked much better overall.
Egypt 2025: TCBZ + Ivermectin Combination (LATEST RESEARCH)
A 2025 study from Egypt examined new treatments. It focused on patients who don’t respond well to standard therapy. Here are the main findings on effectiveness and future treatment direction.
An open-label prospective study at Assiut University in Upper Egypt included 136 patients.
They studied Triclabendazole alone. This was given to 65 patients. They also looked at a mix of Triclabendazole and ivermectin. This combination was given to 71 patients.
Monotherapy had a 26.2% complete response rate. This indicates a lower overall treatment success.
Combination therapy had a 53.3% complete response rate. This shows it is much more effective.
The first clinical strategy aims at triclabendazole treatment failure. Larger trials are needed urgently.
Global Real-World Data (TriNetX 2025)
Data from TriNetX’s global database shows that Triclabendazole is used in only 6% of cases. This data includes 174 patients from 2021 to 2024.
Limited access to the treatment in many regions is likely the reason for this. Despite this, outcomes still stay stable, with 90-day mortality under 6%.
About 10% of patients had biliary complications. These included cholangitis and gallstones.
Safety Profile and Adverse Events
Triclabendazole is generally well-tolerated. It usually doesn’t cause major issues. Most side effects are mild and don’t last long. These side effects typically appear within 1-3 days of treatment.
They’re usually a result of your body’s reaction to parasites dying off, rather than the medication itself being toxic.
Common Adverse Events Table (≥2% incidence, FDA Label)
| Adverse Event | Clinical Note |
|---|---|
| Abdominal pain | 10 mg/kg single/double → 70-100% |
| Hyperhidrosis (sweating) | 10 mg/kg → 85-100% |
| Nausea | 10 mg/kg → 80-100% |
| Decreased appetite | 10 mg/kg → 70-100% |
| Headache | 1, 2 or 3× 10mg/kg → 63.9-68.6% |
| Urticaria (skin rash) | 2× 10 mg/kg → 92% symptom-free at 3 months |
| Diarrhoea / Vomiting | 1 dose: 79.4% cure | 2 doses: 93.9% cure |
| QTc interval prolongation | 2× 10 mg/kg → High efficacy |
| Liver enzyme elevations | 2× 10 mg/kg → High efficacy |
Most side effects are pretty mild and tend to clear up on their own. We should look out for serious effects. These include changes in heart rhythm and higher liver enzymes. This will help to ensure patients can use the treatment safely.
QTc Interval Prolongation – KEY SAFETY WARNING
QTc prolongation is a major safety issue with Triclabendazole. This needs regular monitoring to ensure everything stays on track. The key data below show risk levels, monitoring needs, and drug interactions.

Triclabendazole Resistance – An Emerging Global Concern
Triclabendazole resistance is a major concern for clinicians today. It’s affecting how well treatments work in the long run.
Resistance was first reported in Australian livestock in 1995. Since then, it has appeared in over 11 countries.
Human treatment failures have happened in the Netherlands. They have also been seen in Chile, Peru, Portugal, and Turkey.
New research is adding to this worry. A 2025 study showed that resistance appeared in Peru and the UK. Each location had its own genetic patterns. Pathways like EGFR-PI3K-mTOR-S6K changed.
Microtubules also showed changes. There are thirty genetic markers to detect resistant parasites. This provides around 75% accuracy.
Documented Human Resistance Cases Table
| Country (Year) | Key Finding |
|---|---|
| Netherlands (2012) | First documented human TCBZ resistance – in a farmer |
| Peru – PLOS NTDs (2016) | 7 patients failed multiple TCBZ courses; persistent egg excretion |
| Peru – CDC EID (2021) | 146 children: 55% cure after 1st dose; only 11.6% failed 4 treatments |
| Chile (reported) | Human cases documented – CDC EID 2021 review |
| Portugal (reported) | Human cases documented – CDC EID 2021 review |
| Turkey (reported) | Human cases documented – CDC EID 2021 review |
| UK (2025) | Independent TCBZ-R evolution detected genomically in F. hepatica flukes |
These cases show that resistance is spreading fast in many countries and populations. The problem is getting worse. Cure rates are declining, and treatment failures persist. This shows we need new therapies and better global monitoring.
Clinical Implications of Resistance
Triclabendazole resistance is a big problem. It makes treatment harder. Right now, there is no proven alternative.
Nitazoxanide has shown some effect, but the data are still limited. Artesunate was less effective, with about 76% success in studies. This makes it less reliable than standard treatment.
Some promising findings have emerged from recent research. In 2025, a study in Egypt tested Triclabendazole with ivermectin.
The results were impressive. The response rate was 53.3%. This is much higher than the 26.2% seen with monotherapy.
This is the first tested alternative approach. Large-scale clinical trials are urgently needed to confirm the safety and effectiveness of replacement treatments.
WHO Global Program and Disease Control Data
The World Health Organization manages fascioliasis. It works to control it worldwide. They team up with countries to share drugs and build national programs. Below covers key country initiatives, donation impact, and treatment reach worldwide.
Conclusion: Triclabendazole’s Role in Global Health – An Assessment
Triclabendazole is still a crucial medicine. The World Health Organization has called it the best treatment for fascioliasis.
They have done this since 1997. It’s the only one they suggest for this condition. It has been used for over 25 years and shows cure rates close to 95.5% with a two-dose regimen.
Approved by the U.S. Food and Drug Administration in 2019, it provides an easy one-day treatment. It works at all stages of parasitic infections and has treated about 2 million patients worldwide.
However, rising resistance is now a serious concern. Reports of lower cure rates have been published in multiple countries, including Peru and the UK. Some studies show that the treatment works in only about 55% of children.
New 2025 research shows that combination therapy improves outcomes (53.3% vs 26.2%). This shows we urgently need alternatives, better monitoring, and ongoing global research.
FAQ Section – 8 Key Questions
Q1. What cure rate does Triclabendazole achieve in clinical trials?
Ans:- Clinical reviews show some really strong results over the past 25 years. The usual two-dose treatment gets cure rates of 70-100%.In Egypt, the cure has improved from 79.4% with a single dose to 93.9% with a double dose. Bolivia’s also seen big results from its large programs, cutting prevalence from 5.6% to under 1%.
Q2. Which countries have the highest burden of fascioliasis requiring Triclabendazole?
Ans:- A 2023 meta-analysis reveals that Bolivia has the highest prevalence at 21%. Peru follows with 11%, and Egypt has 6%. South America averages 9%, Africa 4.8%, and Asia 2%. Cases are reported in 81 countries. Over 2.4 million people worldwide are infected.
Q3. Has Triclabendazole resistance been reported in humans?
Ans:- Yes, resistance has been reported. A 2021 CDC study in Peru found that cure rates dropped from 55% after the first treatment to just 23% after four rounds. A separate study found that 74% of patients improved with tweaked treatment plans. This implies that resistance can vary a lot from one person to another.
Q4. What is the FDA-approved dosage for Triclabendazole?
Ans:- The U.S. Food and Drug Administration recommends two doses of 10 mg/kg. Take them 12 hours apart and with food for the best results. For a 70 kg adult, that’s about 700 mg per dose. It’s approved for patients aged 6 and above.
Q5. What are the main side effects according to clinical data?
Ans:- The most common side effect is abdominal pain. It occurs in 56-93% of patients, often from parasite removal. Other effects can include feeling sick, sweating, headaches, and mild changes in liver function. There’s also a rare but serious risk of changes in your QT interval, so some patients will need to be monitored.
Q6. Can I buy Triclabendazole online from India?
Ans:- Yes, Actiza Pharmacy provides Triclabendazole globally. All shipments come from India. It exports to over 25 countries, including the USA, UK, and UAE. Full documents, including the COA, invoice, and certificate of origin, are provided for each shipment.
Q7. Does taking Triclabendazole with food really matter clinically?
Ans:- Yes, it is very important. Taking medicine with food, especially fatty foods, can help the body absorb it better. It’s a smart way to deliver the drug right to the parasites in the bile. This can improve its effectiveness and lower the risk of treatment failure.