Introduction: The High Stakes of Pharmaceutical Supply Chains
The global pharmaceutical market is worth over $1.6 trillion. Its supply chains are very complex and connected. Over 70% of medicines come from multiple countries. This makes the system efficient. However, it is also prone to disruptions.
Unlike other industries, problems in pharma supply chains directly affect patient health. Reliability is therefore extremely important.This is especially true for high-demand therapeutic segments, where businesses depend on trusted partners such as a Fildena Supplier or Cenforce Supplier to ensure consistent product availability across markets.
The COVID-19 pandemic showed how fragile these networks are. It exposed risks. These included relying too much on certain regions. Delays in raw material supply were also a problem. Companies lacked contingency plans.
Now, more regulations and ESG demands are being made. This pushes companies to build supply chains that are transparent, resilient, and ethical.
Market Outlook and Projections (2024–2033)
The industry is starting a decade of growth. This is due to changes in demographics and the need for medical care.
| Metric | Details |
|---|---|
| 2024 Valuation | $1.5 Trillion |
| 2025 Valuation | $1.6 Trillion |
| 2033 Forecast | $2.5 Trillion |
| Growth Rate | 5.8% CAGR (2026–2033) |
Overview of the Global Pharmaceutical Supply Chain
What Is the Pharmaceutical Supply Chain?
The global pharmaceutical supply chain is a carefully organized system. It ensures safe and fast delivery of medications from manufacturers to consumers.
This process is complex and interconnected. It includes several players: suppliers, manufacturers, quality regulators, logistics firms, and healthcare organizations.
They all work together to ensure drugs are available and stay in good condition.
Key Elements of the Pharmaceutical Supply Chain:
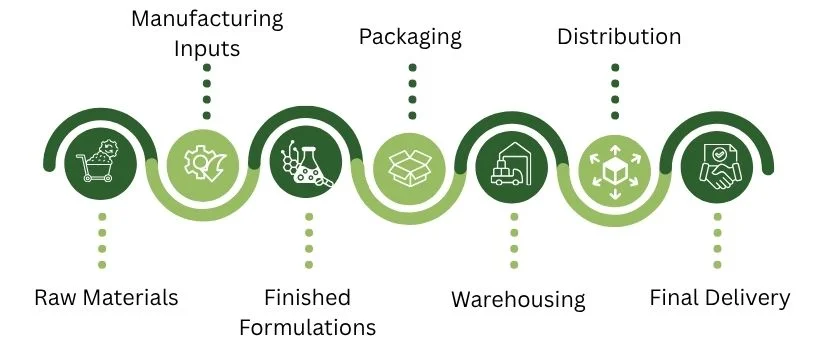
Overview of the global pharmaceutical supply chain
The pharmaceutical supply chain is a complex, cross-border network. It handles everything from raw materials, like APIs and excipients, to manufacturing inputs.
It also distributes finished medications to healthcare providers and patients around the world.
Key Stages of the Supply Chain
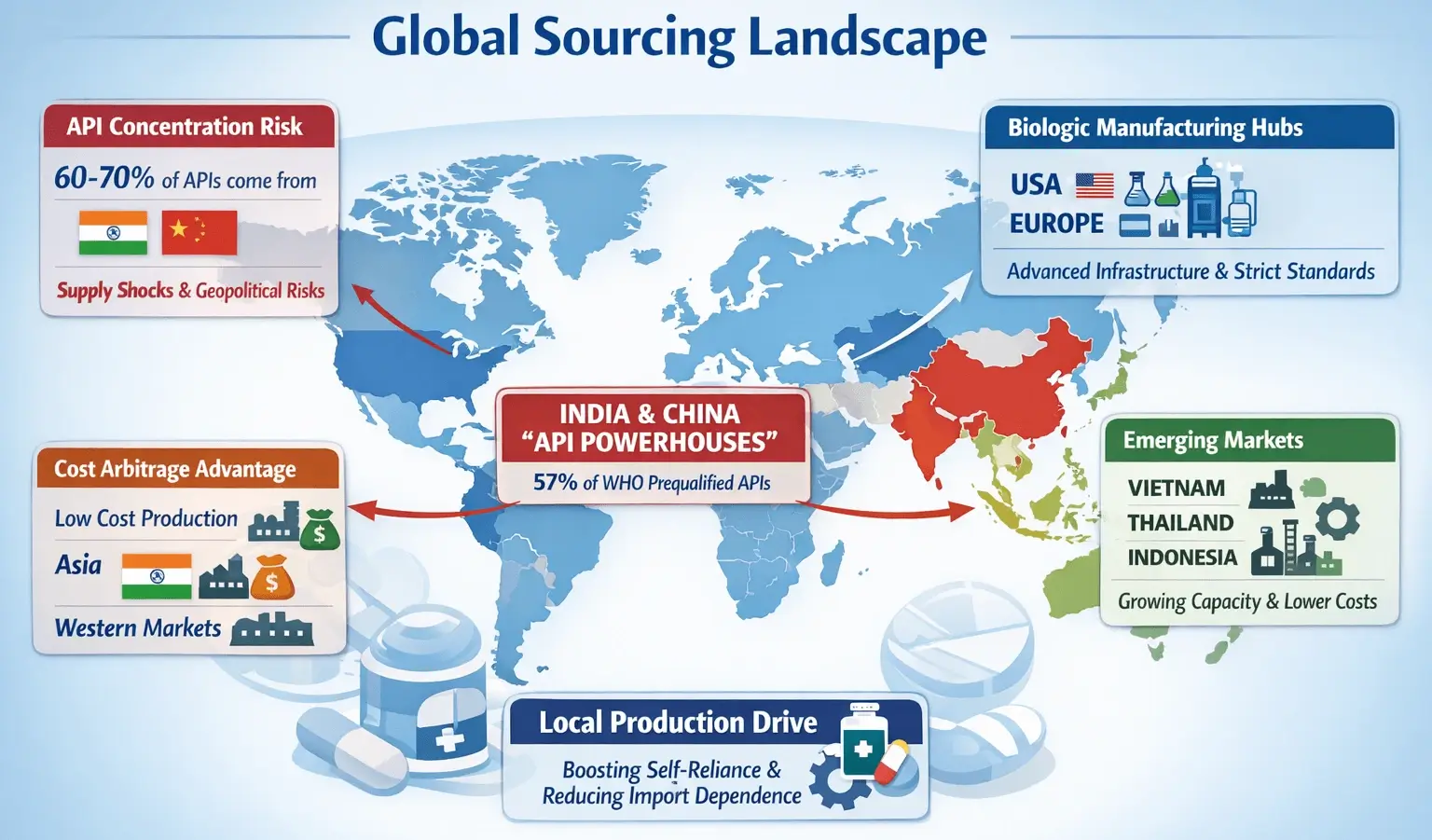
About 60–70% of the world’s Active Pharmaceutical Ingredients (APIs) come from Asia. India and China are top manufacturing hubs. They are cost-effective and can produce goods in large quantities.
India provides about 20% of the world’s generic medicine. This makes it a key supplier of affordable treatments for both developed and developing markets.
Advanced biologic drugs depend on 10 or more specialized suppliers. Each one manages tasks like cell culture, purification, and cold-chain logistics. This setup raises dependency and coordination challenges.
Who Are the Major Players? (And Why It Matters to Buyers)
Many stakeholders influence the pharmaceutical supply chain. Each one controls an important part of the process. Buyers need to know who has influence and where dependencies exist. This can impact sourcing choices, pricing, and supply continuity.
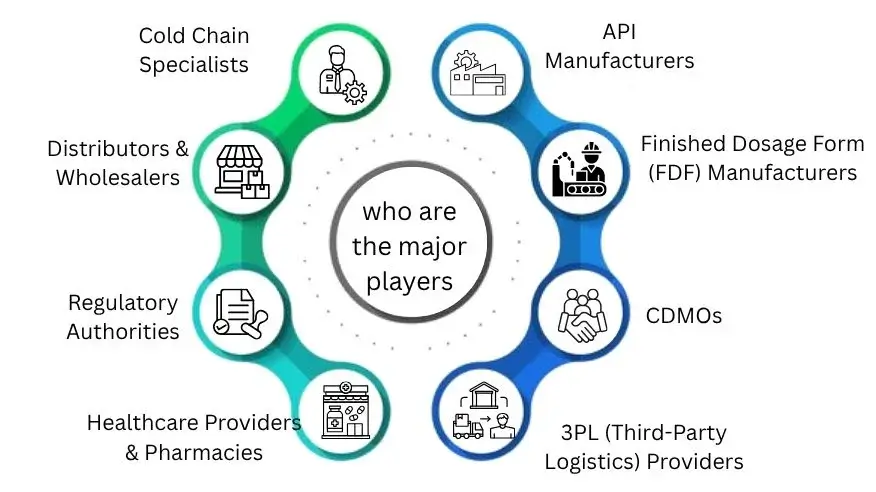
Power Dynamics That Buyers Must Understand:
There is a significant trend toward outsourcing high-value production. In India alone, the CRDMO sector is expected to grow to $14 billion by 2028.
3PL & Cold Chain Specialists:
Third-party logistics are vital now. Over 25% of pharmaceutical products need special “cold chain” handling. This is because they are sensitive to temperature.
Global Sourcing Landscape
Pharmaceutical sourcing today combines cost efficiency, technical skills, and regional expertise. Newer regions are steadily gaining capacity, even as traditional hubs still dominate. This helps reduce global dependency.
About 60–70% of global APIs come from India and China. This brings cost benefits but also risks if there are disruptions in these areas.
The United States and Europe excel in biologics manufacturing. They have advanced infrastructure, strong IP protection, and strict regulatory standards. At the same time, combination drug segments are expanding, increasing the importance of working with a reliable Super P Force Supplier to meet evolving patient needs.
Countries like Vietnam, Thailand, and Indonesia are becoming popular for manufacturing. They offer lower costs and more choices.
These regions are investing in local drug production. This will boost self-reliance and cut down on imports.
API Concentration Risk
Relying too much on a few areas raises the risk of supply shocks, export limits, and geopolitical problems.
Biologic Manufacturing Geography
Biologics mainly thrive in developed markets. They need advanced technology, a skilled workforce, and strict compliance systems.
Cost Arbitrage Advantage
Asia has lower production costs. In contrast, Western markets offer better quality assurance and innovation. This makes buyers weigh cost against reliability.
India & China are the powerhouses for Active Pharmaceutical Ingredients (APIs). India alone manufactures more than 500 distinct APIs and accounts for 57% of the APIs on the WHO prequalified list.
These markets focus on creating high-value biologics, cell therapies, gene therapies, and other advanced treatments.
What Makes Pharmaceutical Supply Chains So Complex?
Cold Chain: Keeping Products Within a Safe Temperature Range
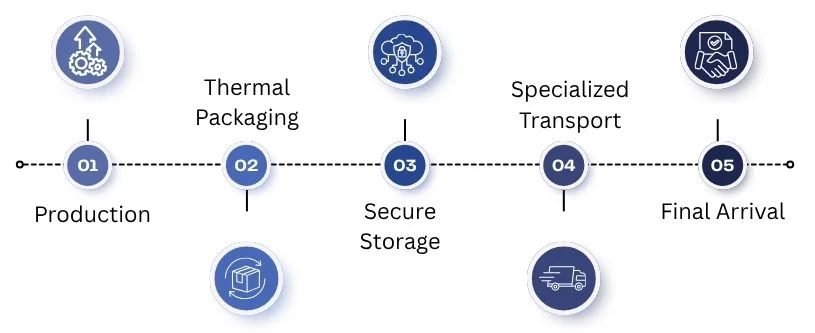
Many modern medicines, like vaccines and biologics, need strict temperature control during transport. Brief exposure outside the required range can harm drug stability. This makes cold chain management a vital and delicate part of the supply chain.
More than 25% of pharmaceutical products need controlled temperatures. This number is rising as biologics become more common.
Biologics typically need storage at 2–8°C. But some advanced therapies need colder temperatures, like -20°C or even -70°C.
Guidelines help with storage, handling, and transport. But, keeping compliance in different regions is tough.
Advanced sensors and tracking systems monitor temperature all the time. They help prevent unnoticed changes.
A cold chain is a logistics network. It uses thermal management to keep perishable items fresh. Maintaining strict temperature ranges prevents spoilage and ensures public health safety.
The 5-Step Logistics Journey
Compliance: Navigating a World of Regulations
Pharmaceutical supply chains follow strict global and regional rules. This keeps products safe and consistent. Standards like GMP, GDP, and GSP control manufacturing, storage, and distribution.
Serialization laws, such as EU FMD and US DSCSA, improve traceability and help stop counterfeiting. Additional requirements, like batch testing and QP release in Europe, create ongoing challenges.
Country-specific labeling adds to the complexity. Compliance is always a localized issue.
Visibility in Pharmaceutical Supply Chains: Knowing Where, What, and When
Modern pharma supply chains need real-time visibility. This helps track product movement and condition at each stage.
End-to-end traceability, backed by serialization data and blockchain, helps stakeholders spot issues fast. This prevents losses and ensures compliance with regulations across borders.
Risk Management: Planning for the Unexpected
Pharmaceutical supply chains need to handle disruptions. These can include pandemics, geopolitical conflicts, API export restrictions, and natural disasters.
Unexpected challenges, such as port congestion and factory shutdowns, can delay vital medicines. So, proactive risk planning and supplier diversification are essential.

Global Reach, Local Knowledge
Pharmaceutical supply chains work worldwide. But their success relies on knowing local rules and market trends.
Every country has its own customs rules, paperwork needs, and laws. This makes it important to partner with local distributors for easy market entry and distribution.
Technology and Innovation: Powering Smarter Pharma Supply Chains
Advanced technologies are changing pharma supply chains. They boost efficiency and accuracy.
AI forecasting, predictive analytics, IoT temperature checks, digital twins, and ERP systems benefit organizations. They can anticipate demand, cut waste, and make better decisions in real time.
Sustainability: Meeting Environmental Goals Without Compromising Safety
Sustainability is now a top priority in pharmaceutical logistics. Companies are focusing on cutting carbon emissions and meeting ESG standards.
Eco-friendly packaging, better transportation, and reverse logistics for waste are helping companies. They can now meet safety standards while being environmentally responsible.
Operational Proficiency in the Pharmaceutical Supply Chain
Warehousing and Inventory Management
Pharma warehousing focuses on precision and product safety rather than just storage. FEFO (First Expiry, First Out) uses near-expiry medicines first.
This cuts down on waste. Also, safety stock planning stops shortages without overloading inventory.
Advanced expiry tracking systems and multi-warehouse strategies keep products available. They also speed up distribution across different regions.
Supply Chain Visibility and Traceability
Visibility in pharma supply chains is driven by the ability to track every unit in real time. Track-and-trace systems, backed by serialization, allow complete product identification and movement history. These systems boost transparency when linked to regulatory reporting. They help with compliance and allow quick responses to recalls or disruptions.
ERP in Pharma Supply Chain
ERP systems play a crucial role in pharmaceutical supply chains. They link procurement, manufacturing, inventory, quality control, and distribution into one unified platform. ERP enables real-time data flow and automates processes.
This helps companies stay compliant, cut down on errors, and make better decisions. In the pharma industry, which has strict rules and tight deadlines, ERP improves coordination, traceability, and efficiency in the supply chain.
What Is ERP in Pharma?
ERP in the pharmaceutical industry is software.
It connects planning, operations, and compliance management. It brings everything together in one system. It ties together procurement, manufacturing, quality control, and distribution.
This ensures regulatory requirements are consistently met. Centralized data and standardized processes help everything run efficiently.
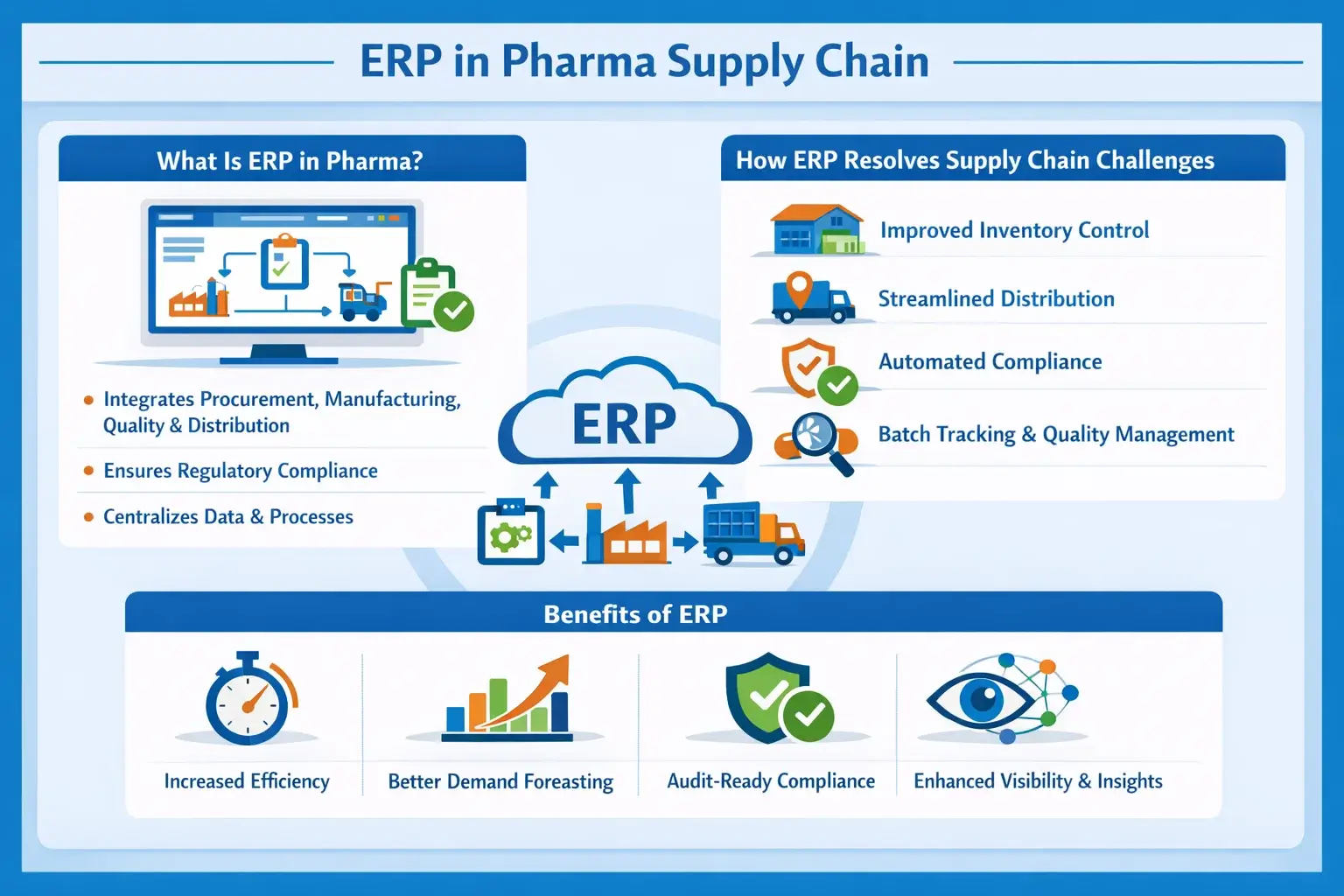
How ERP Resolves Supply Chain Challenges
ERP systems address key supply chain issues by creating a centralized flow of accurate, real-time data.
They enhance inventory control, simplify distribution planning, and automate compliance tasks.
This cuts down on manual errors. Also, features like batch traceability and quality management help track products better. This means faster recalls and consistent standards.
Benefits of ERP
ERP boosts efficiency by streamlining operations.
It also reduces delays throughout the supply chain. It cuts down on stock-outs by improving demand forecasting.
Plus, it keeps companies audit-ready with built-in compliance tools. Overall, it enhances transparency, allowing better visibility and smarter decision-making at every level.
Regulatory Environment & Quality Control
Pharma supply chains are tightly regulated to ensure drug safety and quality.
Authorities like the FDA, EMA, CDSCO, and TGA enforce rules such as import alerts, batch certification, and testing.
The WHO Prequalification Programme and pharmacovigilance systems keep a close eye on drugs, even after they are sold.
Cost Drivers & Price Transparency
Multiple cost layers across the supply chain influence pharmaceutical pricing. APIs account for the largest share (40–60%), followed by manufacturing, quality checks, and packaging.
Cold chain logistics, insurance, and import duties add extra expenses. This affects the final cost. So, transparency is key for making accurate pricing decisions.
Supply Chain Disruptions – Lessons from COVID-19
The COVID-19 crisis showed serious problems in pharmaceutical supply chains.
Export bans and soaring air freight prices suddenly interrupted the global supply of medicine.
Relying too much on a few API sources caused shortages. Also, poor inventory planning led to stockouts and overstocking.
These challenges emphasized the need for diversification, better forecasting, and stronger supply strategies.
Why a Broader Supplier Ecosystem Matters
Using a larger supplier network helps pharma companies lower risk and keep supplies steady.
A diverse ecosystem reduces reliance on one source. This helps businesses react quickly to disruptions, price changes, and shifts in demand. It also ensures steady product availability.
Best Practices for Buyers
Effective pharmaceutical sourcing requires a structured approach that balances quality, compliance, and reliability. Buyers should look past just pricing.
They need to assess suppliers on their operational strength, regulatory history, and risk readiness. This helps ensure stable supply in the long run.
Supplier Evaluation Framework
Effective pharmaceutical sourcing requires a structured approach that balances quality, compliance, and reliability. Buyers should look past just pricing.
They need to assess suppliers on their operational strength, regulatory history, and risk readiness. This helps ensure stable supply in the long run.
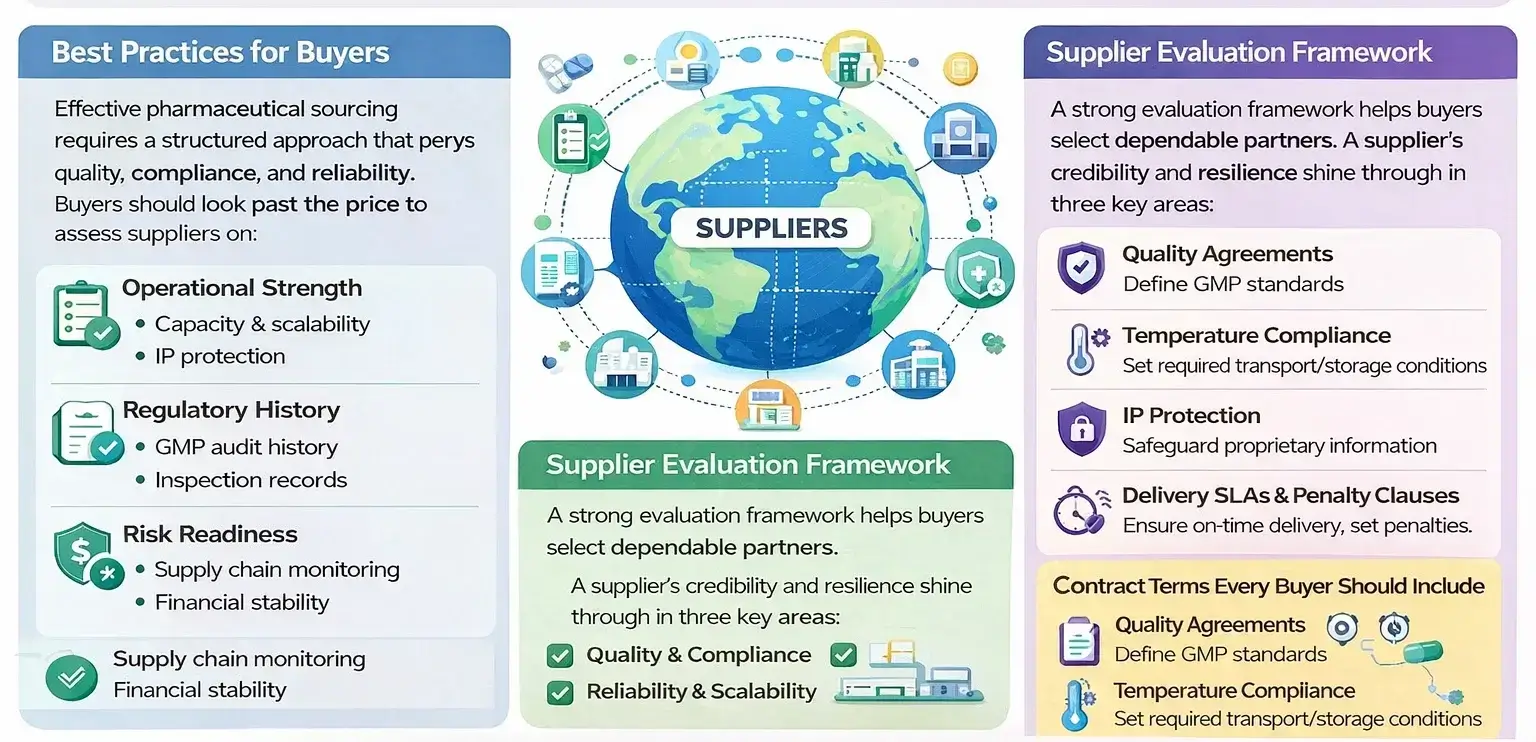
Contract Terms Every Buyer Should Include
A strong evaluation framework helps buyers select dependable partners.
GMP audit history and inspection records show compliance reliability. Also, financial stability helps ensure continuity.
A supplier’s credibility and resilience shine through in three key areas:
Supply Chain Monitoring & KPIs
Well-defined contracts protect buyers from operational and legal risks. Including quality agreements, temperature compliance clauses, and clear recall responsibilities ensures accountability.
IP protection keeps proprietary information safe. Delivery SLAs and penalty clauses ensure performance standards are met. They also help reduce disruptions.
The Future of Pharmaceutical Supply Chains
Pharma supply chains are moving to a hybrid model. This change balances global efficiency and regional resilience.
AI and blockchain are boosting prediction, tracking, and trust. Also, reshoring in the US and EU cuts down dependency risks.
Smart packaging, automated warehousing, and ESG-focused sourcing are making supply networks more agile and responsible.
Conclusion: The Patient Benefits
Pharmaceutical supply chains ultimately serve as a backbone for patient safety. Transparency strengthens trust, compliance reduces disruptions, and technology improves visibility across every stage.
By diversifying sources and making smarter buying choices, the system gains reliability. This ensures steady access to safe and effective medicines.
