Introduction: The Expanding Global Pharmaceutical Trade Landscape
The global pharmaceutical market is set to hit about $1.6 trillion by 2025. This shows a big rise in healthcare demand around the world.
It’s a big deal because more people around the world need healthcare services. Generic medicines are important in this system.
They are bioequivalent to branded drugs but cost 80-85% less. This helps millions of patients get affordable treatment.
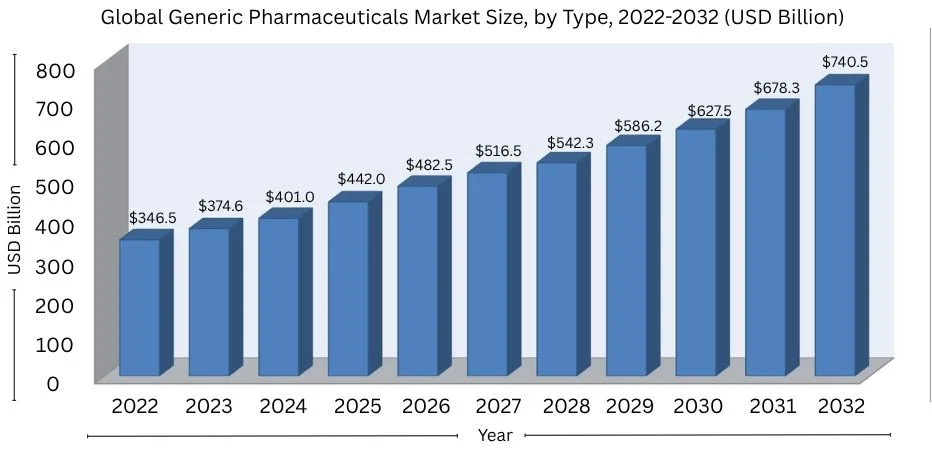
In 2022, the global market for generic drugs was worth USD 346.5 billion. It is set to hit USD 740.5 billion by 2032.
This represents a growth rate of 8.1% per year. There’s a rising need because of more chronic diseases, aging populations, and global trade growth.
A clear sourcing strategy is key for international buyers. It’s also important to work with trustworthy suppliers.
This really helps keep standards and follow regulations. It also protects against a major issue: the $200 billion counterfeit medicine trade.
Market Drivers
Global healthcare trends are driving up the need for generic medicines. This is happening in developed markets. It’s also happening in emerging markets. It is a significant shift.
Buyer Impact
The global generics market is seeing some growth. Global pharmaceutical buyers will face new challenges. However, they’ll also have more chances to find the right suppliers.
Regulatory Frameworks & Compliance in Destination Markets
National regulatory authorities check that medicines are safe. They also make sure the medicines are of high quality and handle approvals for global trade. This key role ensures our medicines are safe and effective.
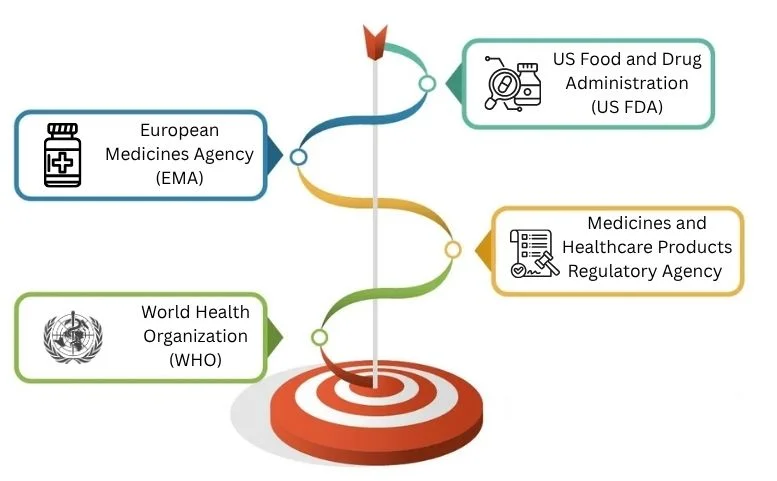
Major Global Regulatory Authorities
Global regulators set standards. They oversee inspections and give approval for products in major pharmaceutical markets. They’re in charge of making sure everything meets the right standards.
Key Compliance Requirements
Pharmaceutical exporters have strict rules. They need documents and approvals to sell medicines abroad.
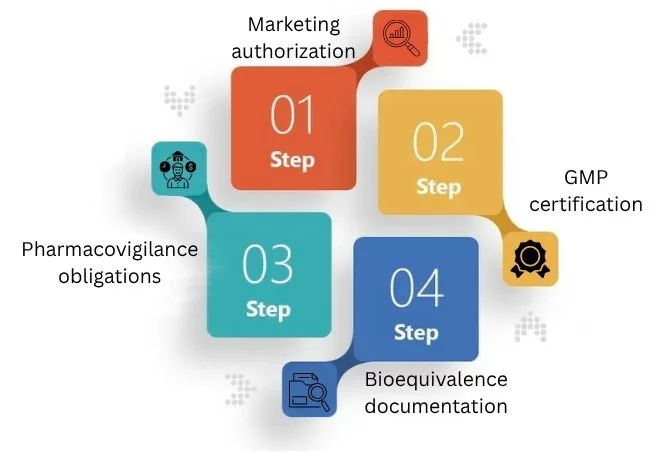
Below outlines the core compliance elements for companies:
Regulatory Risk & Penalties
Not meeting regulatory requirements can lead to issues. This affects the global pharmaceutical trade. It’s simple: if you don’t comply, you’ll face big problems.
Authorities have the power to issue import bans or Import Alerts.
If they do, they can seize products and hit companies with fines worth up to 100% of the shipment’s value. It can really hurt a company’s reputation.
Step 1 – Define Sourcing Strategy
A good sourcing strategy is very important for pharmaceutical buyers. It helps them find reliable partners and makes sure they have a consistent supply of products.
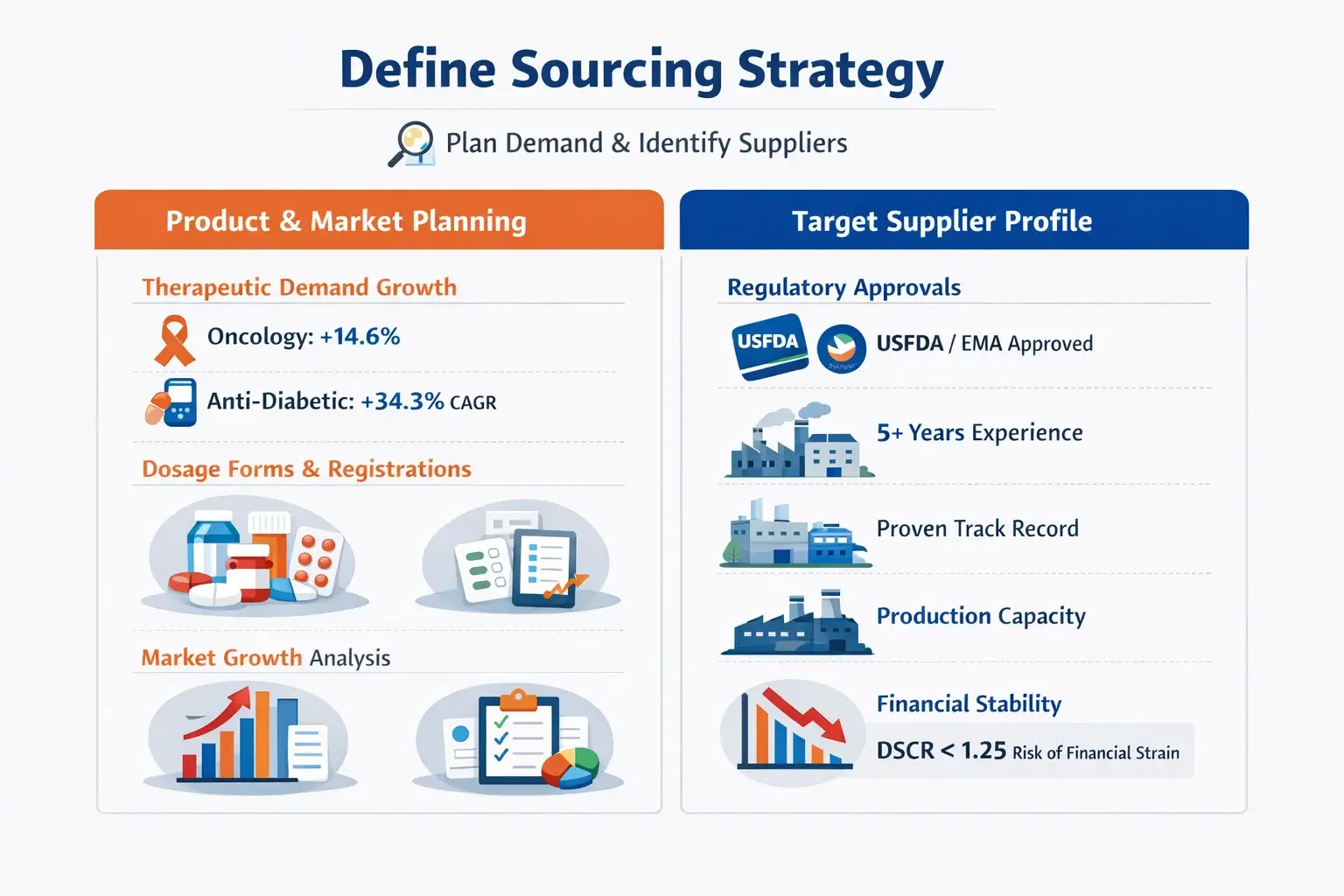
Below explains how to plan product demand and identify suppliers:
Product & Market Planning
Buyers need to plan their sourcing. They’ve got to look at therapeutic demand, the different dosage forms, and market growth.
High-growth areas are oncology and anti-diabetic medicines. Oncology has a 14.6% growth rate. Anti-diabetic medicines have a 34.3% CAGR. These require strong volume forecasts.
They also need proper dosage forms and clear product registration.
Target Supplier Profile
A top pharmaceutical supplier must have the right approvals. These approvals ensure they meet all regulations.
They should also show a strong history of successful exports. Partners with USFDA or EMA approval are key. They should have at least five years of operation.
They need enough production capacity. A Debt Service Coverage Ratio below 1.25 may show financial risk.
Step 2 – Supplier Identification & Screening
To find the right pharmaceutical suppliers, do some research. Then, screen them closely. This way, you can make smart partnership decisions.
The following explains how buyers identify suppliers:
Data-Driven Sourcing Channels
In pharmaceutical sourcing, accuracy and reliability are key. Using verified databases is a great start. Also, attend global trade events to connect with professionals in your network.
They help companies make smart choices and stay competitive. Here are the main platforms buyers use to find trusted manufacturers and suppliers around the globe.

Screening Criteria
Buyers need to check suppliers’ regulatory history. They should also look at risk factors. This should happen before making a shortlist.
Below outlines critical compliance factors:
Quality monitoring also uses a metric called the Official Action Indicated. In 2025, India’s OAI cases dropped from 20 to 11. This shows that compliance standards have improved.
Shortlisting Framework (Scoring Model)
A structured scoring system helps buyers compare suppliers fairly. It also reduces the risks in sourcing.
Here are the main factors to consider when creating a weighted supplier selection model:
Step 3 – Technical & Regulatory Due Diligence
This step checks if products meet safety, quality, and legal rules before approval. It explains documents, patent checks, and supply chain risks.
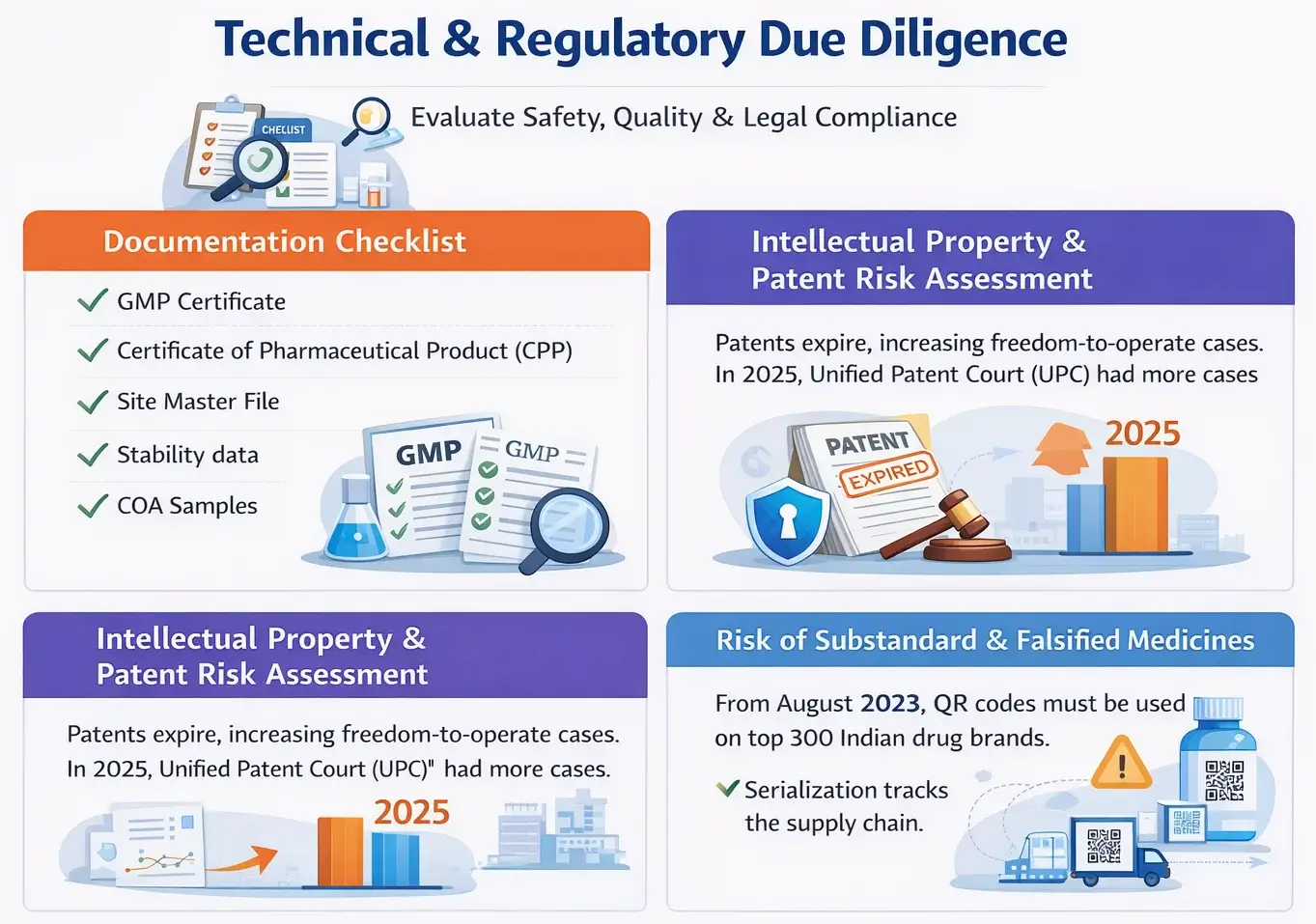
Documentation Checklist
Here are the main documents to check product quality, safety, and compliance.
Intellectual Property & Patent Risk Assessment
Patent checks are essential to avoid legal issues before launching a product. First, check if any patents have expired.
Then, confirm the product is clear to sell. A freedom-to-operate review is also crucial and must be done thoroughly. In 2025, the United Patent Court (UPC) had more cases.
These cases were about patent infringement and generic drug launches.
Risk of Substandard & Falsified Medicines
Supply chain checks are essential to lower the risk of fake or low-quality medicines.
Be clear about your suppliers and carefully vet all sources. Serialization is essential.
From August 2023, QR codes must be used. This applies to the top 300 Indian drug brands. They will help verify authenticity.
Step 4 – Supplier Evaluation & Audits
This step checks if suppliers meet quality standards. It also ensures they follow safety standards before approval. It covers document reviews, site audits, and inspections by third parties.
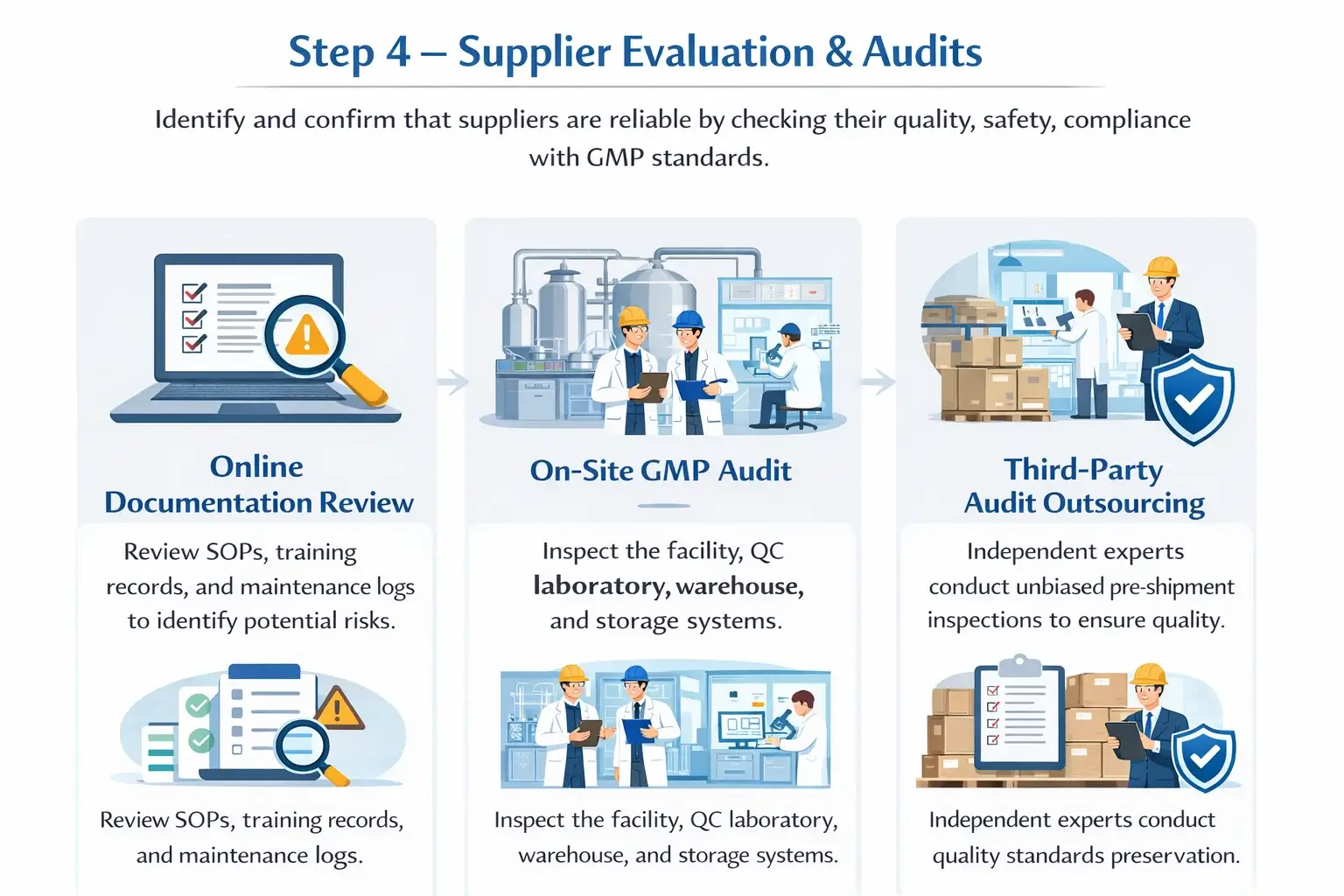
Online Documentation Review
It checks supplier documents to spot potential risks quickly. It reviews standard operating procedures. It also checks training records and maintenance logs. The aim is to ensure that their systems are effective.
On-Site GMP Audit
On-site audits confirm real conditions at the supplier facility.
This includes inspecting the manufacturing facility. It also means reviewing the QC laboratory and checking the warehouse and storage systems, too. This helps with proper handling, keeps the right temperature, and ensures traceability.
Third-Party Audit Outsourcing
Independent experts help provide an unbiased look at suppliers. Third-party GMP audit firms perform thorough checks.
Pre-shipment inspection makes sure products meet quality standards before shipping. This reduces risk in the supply chain.
Step 5 – True Landed Cost Analysis & Commercial Negotiation
This step calculates the total cost from the factory to delivery. This helps in making better decisions. It covers cost factors, modeling methods, and negotiation strategies.
Beyond Ex-Factory Pricing
Focusing only on the ex-factory price can be misleading. It doesn’t include extra costs, which can add 15-35% to the total cost.
Include freight, insurance, customs duties, and port and warehousing charges. This will give you a clearer view of the product’s cost.

Total Landed Cost Modeling
Cost modeling helps you track expenses and profit in a clear way. It considers how currency changes, payment terms, and cash flow affect your business.
Optimizing MOQ helps balance unit costs with inventory risks. This way, you can make informed choices.
Data-Driven Negotiation Tactics
Having the full cost data on hand really helps when negotiating with suppliers. You can save money by using bulk buying power, bundling products, and signing long-term deals. This also helps you secure stable supply terms.
Step 6 – Logistics & Supply Chain Resilience
This step is key to getting pharmaceutical products to global markets safely and on schedule.
Transport Mode Selection
This section explains how to choose the right transport method and plan delivery timelines. Here are the key differences between shipping options and their impact.
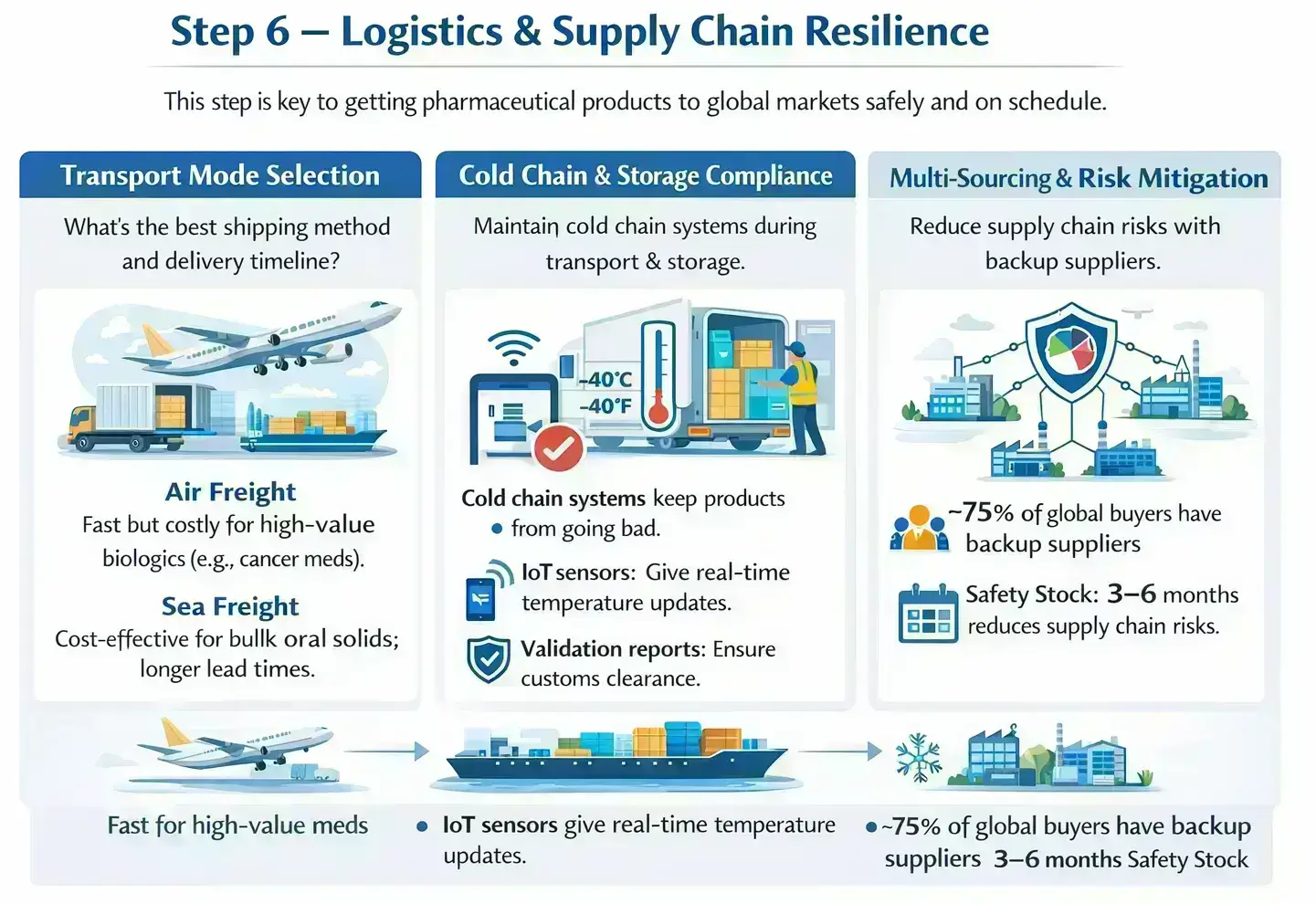
Cold Chain & Storage Compliance
Cold chain systems keep products from going bad. They maintain quality when goods are on the move or in storage.
These systems are crucial for biologics.
They rely on IoT-enabled sensors to remain effective. They provide real-time temperature updates. They also generate validation reports needed for customs clearance.
Multi-Sourcing & Risk Mitigation
Using multiple suppliers reduces supply chain risks. It also helps you avoid depending too much on one company. Most global buyers, about 75%, have backup suppliers in place. A safety stock that lasts 3-6 months keeps operations smooth during disruptions.
Step 7 – Documentation & Customs Clearance
This step ensures products enter the market smoothly. It follows customs rules and gets the paperwork right.
Required Import Documentation
This section shows the key documents for customs approval. It also covers what you need to clear shipments. It explains the paperwork needed for imports to comply with regulations.

Clearance Timelines & Common Delays
This section shows common problems. These issues slow down customs clearance. Below are the required documents to avoid key risks that can delay shipment approval.
Wrong HS classification is a big reason for delays. The code that gets the most attention is HS 3004, which covers both brand-name and generic drugs.
Step 8 – Quality Assurance & Batch Release
This step makes sure every batch is up to standard for quality, safety, and regulations before it’s used. It covers checks, storage, and what to do if something goes wrong.
Incoming Inspection
This section checks product quality when shipments arrive at the facility.
Inspection steps and verification processes include:
Each imported batch must get certified. A Qualified Person (QP) or the Head of Quality will ensure it meets EU/UK standards.
Stability & Storage Compliance
Proper storage is key to keeping your products in good condition over time. To avoid losses, watch the temperature closely. Also, use the First Expired, First Out system. This way, you’ll reduce waste and make sure products get used on time.
Corrective & Preventive Actions (CAPA)
CAPA systems are key in resolving quality issues and preventing future problems. They handle complaints and prepare for recalls. A company with major weaknesses, such as OAI, needs a solid CAPA system. Without it, the company could face issues and might get warning letters.
Step 9 – Post-Shipment Performance Monitoring
This step is about tracking how suppliers perform after delivery. It’s all about quality and reliability. Here are the key metrics and methods for evaluating suppliers.
KPIs to Monitor
This section shows key indicators. They measure how reliable and quality suppliers are. This shows the key metrics tracked after shipping.
Wholesalers should aim to deliver on time over 95% of the time and keep their batch rejection rates under 2%. This helps them perform consistently.
Supplier Scorecards & Dashboards
Supplier scorecards track performance over time. They use simple data to help. Checking progress every quarter helps. Tracking compliance trends also supports decision-making. It’s important to watch how fast suppliers respond. The aim is to reply in under four hours.
Financial Risk & Payment Security
Here are secure payment methods, risk tools, and protection strategies:
Letter of Credit
A Letter of Credit (LC) is a payment guarantee from a bank. It helps reduce risk when dealing with unknown parties. It makes sure sellers get paid on time. This matters in cross-border trade. Trust issues and delays often occur.
Advance Safeguards
Advance payment safeguards offer buyers some security. These safeguards keep them safe. They help avoid risks like not receiving their goods. They also ensure they don’t end up with low-quality items. To do this, they use tools like escrow accounts, standby letters of credit, milestone payments, and guarantees. These tools help them get their money back if suppliers don’t deliver.
Credit Insurance
Credit insurance protects businesses. It covers them if buyers don’t pay or are late with payments. It can cover up to 90% of losses. It helps recover debts and boosts cash flow stability in uncertain markets.
Currency Hedging
Currency hedging cuts losses. It protects against changes in exchange rates in global trade. It’s very helpful. Tools like forward contracts and options help you lock in prices. This helps to protect margins and plan your finances more steadily.
High-Growth Therapeutic Segments (2026 Outlook)
Here’s a brief overview of key segments and growth trends expected in 2026. These therapy areas are growing fast because of more diseases and higher demand.
Oncology generics
Generic cancer drugs are growing quickly. They are expected to increase by 11.3% to 14.6%. This rise is due to more cancer cases.
Cardiovascular medicines
Cardiovascular generics make up the biggest share at 22%. This is due to an ageing population and lifestyle diseases.
Anti-diabetic therapies
Anti-diabetic drugs are growing fast. GLP-1 agonists are set to rise at a 34.3% CAGR.
Anti-infectives
Anti-infectives are in steady demand. They grew by about 7.6% in value. This growth comes from infections and seasonal illnesses.
CNS products
CNS drugs are on the rise. This is thanks to increased awareness of mental health. There’s strong global demand and steady market growth.
Why India Remains a Strategic Long-Term Partner
India holds a strong spot in the global pharma supply chain. Its quality and scale are hard to match. The country has strong regulations and business advantages.
India’s Regulatory Ecosystem
India has a strong regulatory system that is improving. It helps the country meet global quality standards. This system has key authorities. It includes global approvals and strong compliance.

CDSCO Authority
The Central Drugs Standard Control Organization handles approvals and checks the quality. They have recently set up a new online system for Good Manufacturing Practice. This system started in March 2025.
USFDA Plants
India has more than 670 plants approved by the USFDA. This is the highest total outside the US. This helps the country build a strong global reputation. It also helps meet the required standards.
WHO Facilities
In India, WHO-prequalified facilities make high-quality medicines. They supply these medicines around the world. These facilities meet strict safety and effectiveness standards.
Competitive Advantages
India gives big cost savings for global pharma companies. It also offers scale advantages and supply benefits.
Below are the competitive advantages of India over other nations:

API Strength
India’s got a strong API ecosystem. Lots of manufacturers are on board, which means a steady supply and less reliance on imports.
Low Cost
Production costs in India are 30-70% lower. This is compared to Western countries. It helps make medicines cheap for everyone.
Export Scale
India has a strong export capacity. This is backed by trade benefits, like the EU-India FTA, which removes 11% tariffs. There are also incentives available until March 2026.
Red Flags International Buyers Should Avoid
Look out for signs that a supplier might be risky or unreliable.
Buyers should spot these red flags before sealing any deal. This helps to prevent issues in the future.
Low Pricing
Watch out for prices that are too low. You might get a bad deal with poor quality, fake materials, or extra costs.
Always verify prices that are much lower than market rates before making decisions.
Missing Data
If a supplier has no stability data, they can’t prove their product lasts.
Without this data, it’s unclear how the product holds up over time. The shelf life and safety of these products depend on proper testing. If testing is missing, it poses a serious risk.
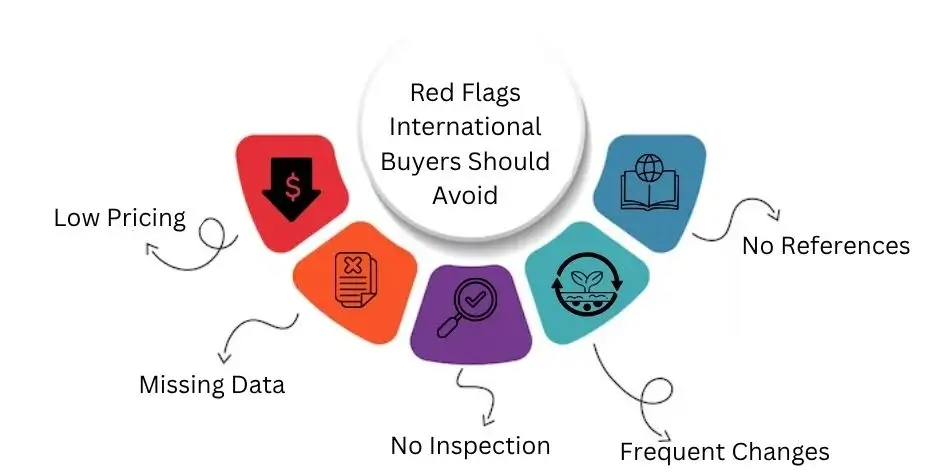
No Inspection
A company with no regulatory inspections likely has weak compliance.
It probably also has low-quality systems. Trusted suppliers often undergo audits from groups like the FDA or ISO. This shows they are credible and meet specific standards.
Frequent Changes
Many name changes can be a warning sign. They might indicate the company is hiding a bad reputation, legal issues, or unhappy customers.
A clear and steady business identity is vital. It helps build trust over time.
No References
If a supplier can’t give you export references, they may have less global experience.
This might mean a higher risk. Find suppliers who can give shipment records and client references. This will help prove their claims. It shows they are reliable and quality-focused.
Strategic Conclusion
Generic sourcing isn’t just about buying cheaply. It’s a procurement process that is managed well to reduce risks. In a fast-growing market, compliance is more important than cost.
Poor quality or failing to meet regulations can cause big losses and rejected shipments.
A solid due diligence approach can reduce import risks and boost long-term success. It helps cut down on problems. This keeps operations smooth over time.
Strong supplier relationships help build trust. Continuous monitoring and a focus on compliance are key. These steps improve supply stability. They also drive profits in the global pharmaceutical trade.
