Sorry, nothing in cart.
Introduction
Generic medicines are the backbone of modern healthcare systems, helping patients access safe and effective treatments at much lower prices.
They help people get safe and effective drugs for much less than the original price.
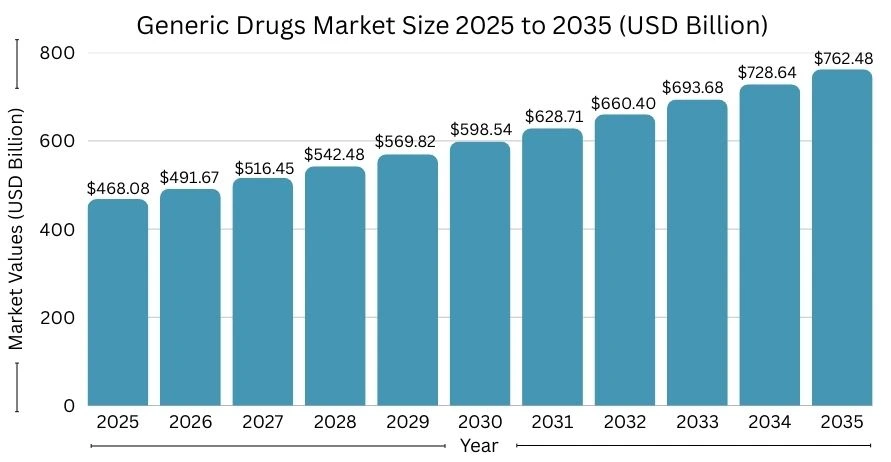
The global market for generic drugs is already huge. One study estimates that the market size is about USD 468 billion in 2025.
It is expected to grow to around USD 762 billion by 2035, which is a growth of about 5% per year.
Another report gives a similar picture. It puts the market at about USD 426.3 billion in 2025 and shows strong growth through 2030 and beyond.
Why is this market so strong? In many large countries, generic medicines make up almost 90% of prescriptions. They are the most common choice for patients.
At the same time, they take up only a smaller share of total spending on medicines. In simple words, generics do most of the work but cost much less.
India has a special place in this story. The country is one of the largest suppliers of generic medicines in the world.
It provides about one-fifth of the global volume of generic drugs and a large share of vaccines. Because of this, India is often called the “Pharmacy of the World.”
Understanding Generic Medicines
Generic medicines are versions of original branded medicines. They come to the market after the patent on the original drug has expired. A generic medicine has the same active ingredient, strength, dosage form, and way of taking it as the brand-name product.
Difference between branded and generic drugs
Branded drugs are developed and first sold by the innovator company. That company spends large sums on research, clinical trials, and marketing.
Generic drugs appear later, once legal protection ends. They do not repeat the basic research so that they can be sold at a lower price.
The main differences are the name on the pack and the cost to the buyer. The medical effect, when the generic is approved, is expected to be the same.
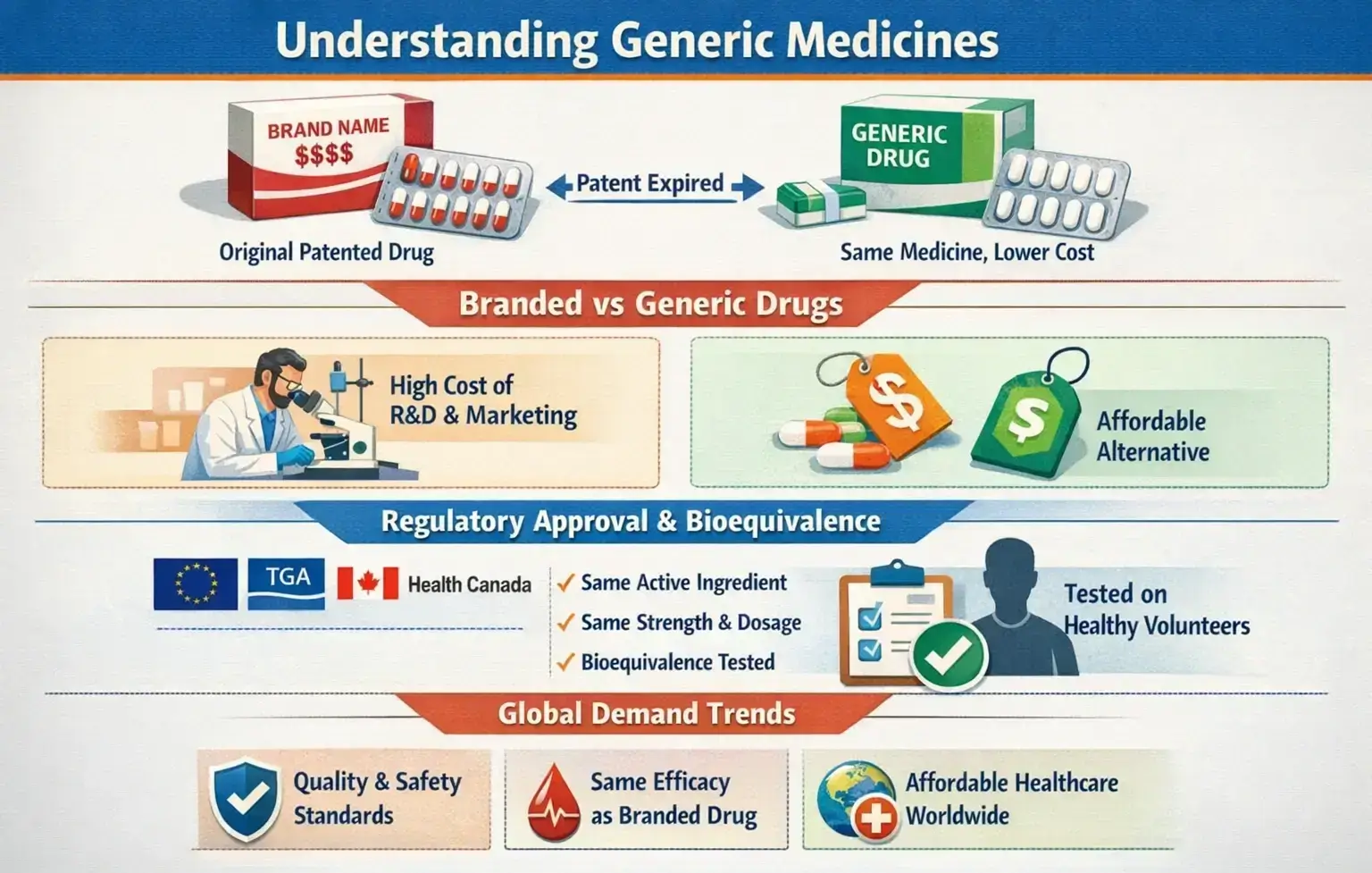
Regulatory approval and bioequivalence
Indian manufacturers have built strong regulatory credentials. They are launched in the market after the original drug’s patent has run out.
A generic drug has the same active ingredient, strength, form, and method of administration as the original drug.
Many plants hold EU-GMP, TGA, Health Canada, and other approvals. This gives buyers confidence that Indian suppliers understand and meet global standards.
Global Demand Trends
A generic drug must pass strict checks before it can be sold. To gain approval, the manufacturer has to prove that:
These tests are called bioequivalence studies. They are usually done on healthy volunteers under controlled conditions.
India’s Role in the Global Generic Market
India is one of the top suppliers of generic drugs in the world. India contributes around 20% of the global volume of generic drug exports and 60% of the total vaccine supply in the world. India is ranked in the top three places in the world in the volume of pharmaceutical products manufactured in the country.
India’s pharmaceutical industry has 3,000 drug companies and 10,500 units. It also includes many chemists, pharmacists, and engineers.
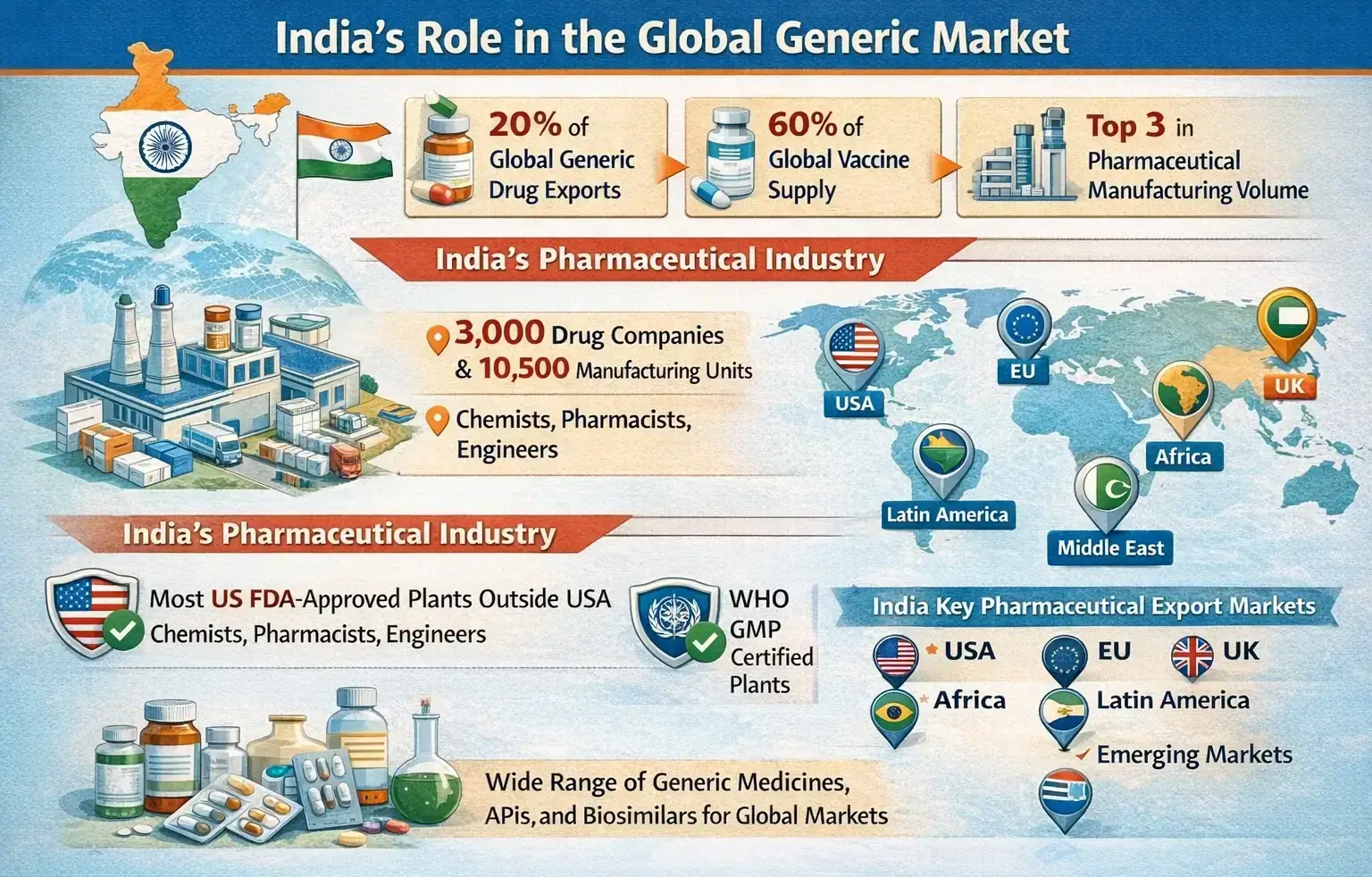
India also has the most US FDA-approved manufacturing plants outside the US and WHO GMP-certified plants.
India’s key pharmaceutical export markets are the US, EU, UK, Africa, Latin America, the Middle East, and various emerging markets.
Several leading Indian pharmaceutical manufacturers contribute to the country’s strong export ecosystem, producing a wide range of generic medicines, APIs, and biosimilars for global markets.
A Snapshot of Pharmaceutical Exports from India
Recent figures show how fast India’s pharmaceutical exports are growing. Exports reached about USD 25.4 billion in 2023 and continue to rise. For the financial year 2024-25, exports are reported at around USD 30.47 billion, which means an annual growth rate close to 9.4%.
India exports medicines to over 200 countries. These include highly regulated markets such as the United States, the United Kingdom, and the European Union, and also many middle-income and low-income countries.

The main export categories are:
This mix of products means India can support both bulk public-health programmes and niche commercial launches.
Key Benefits of Sourcing Generic Medicines from India
When global companies look for supply partners, India stands out for clear, data-based reasons.
Cost Advantage
Manufacturing generics in India is typically 30-40% cheaper than making the same products in the US or EU.
The reasons include lower labour, energy, and land costs. Government support, such as Production Linked Incentive (PLI) schemes and dedicated pharma parks, lowers costs further.
This allows Indian exporters to offer strong prices without cutting corners on quality.
Strong Manufacturing Infrastructure
India has over 3,000 pharmaceutical companies and more than 10,500 manufacturing plants. This creates one of the largest pharma footprints in the world. Multiple sites per company allow flexible scheduling, backup capacity, and support for very large orders.
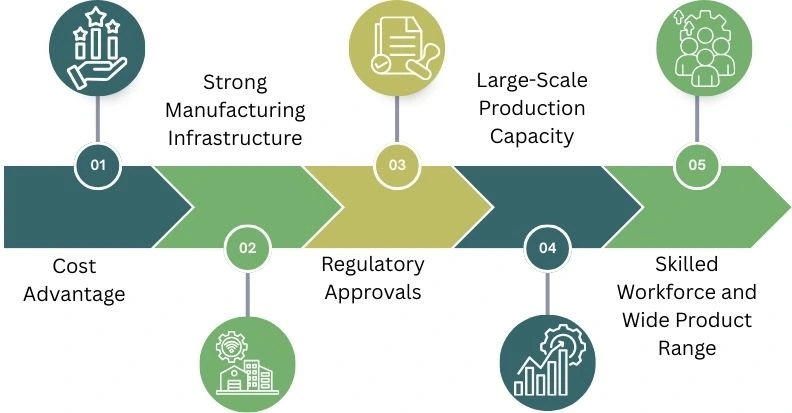
Regulatory Approvals
India’s industry has deep experience in global regulation. There are more than 2,000 WHO-GMP-certified plants in the country. India also has the largest number of US FDA-inspected facilities outside the US.
Many plants hold EU-GMP, TGA, Health Canada, and other approvals. This gives buyers confidence that Indian suppliers understand and meet high regulatory standards.
Large-Scale Production Capacity
With thousands of plants and strong process know-how, Indian exporters can support large commercial batches, supply to many markets at once, and handle long product lists. High capacity helps keep unit costs low and supports a stable supply.
Skilled Workforce and Wide Product Range
India’s pharma workforce includes a large pool of chemists, pharmacists, and engineers experienced in generics and APIs.
The country produces nearly all major therapeutic classes, from anti-infectives and cardiovasculars to oncology, diabetes, CNS, respiratory, and advanced products such as biosimilars and complex injectables.
This breadth gives buyers flexibility to build or expand a full generic portfolio with a small number of partners.
Step-by-Step Process to Source Generic Medicines from India
Define Product Requirements
Begin with a clear internal brief.

Set down:
A clear brief makes it easier to filter suppliers and request accurate offers.
Identify Reliable Suppliers

Use several channels:
Shortlist suppliers based on product range, approvals, capacity, and reputation in the market.
Verify Certifications and Compliances

For each shortlisted supplier, request and review:
This step is vital for reducing risk, especially if you supply highly regulated markets.
Requesting Documentation

Once you narrow the list, ask for key technical documents:
Check that product specifications match the pharmacopoeia (such as USP, EP, BP, IP) or your own in-house standard.
Negotiate Pricing & MOQ

Discuss and align on:
When comparing offers, factor in not just the ex-factory price but also the cost of regulatory filings, local testing, and logistics.
Sample Testing & Quality Check

Before signing a long-term contract:
Record all results and observations. These will feed into the final quality agreement.
Finalize Contracts & Agreements
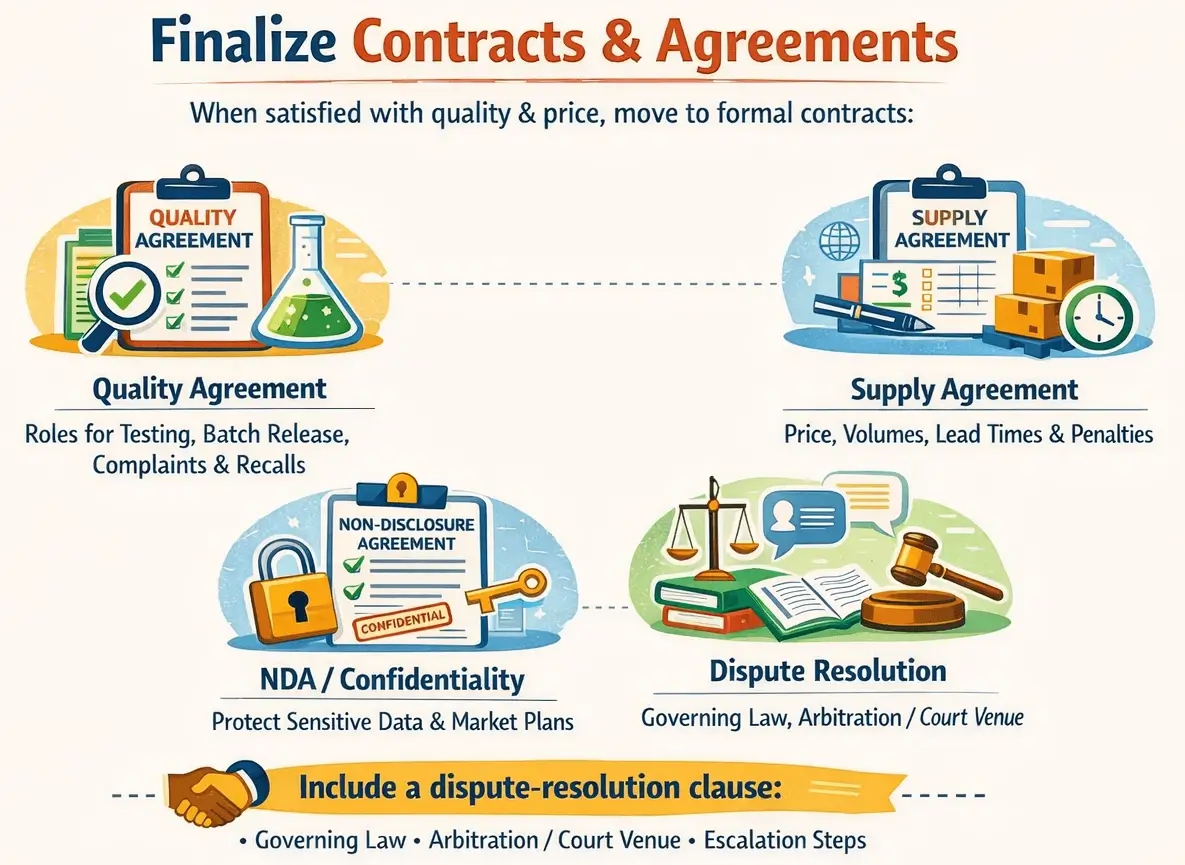
When you are satisfied with both quality and price, move to formal contracts. This includes a Quality Agreement that sets roles for testing, batch release, change notification, complaints, and recalls.
You should also establish a Supply Agreement covering price, volumes, lead times, safety stock, and penalties for non-performance.
In addition, sign an NDA/Confidentiality Agreement for sharing sensitive data and market plans.
Finally, include a dispute-resolution clause that defines the governing law, arbitration or court venue, and escalation steps.
Regulatory & Compliance Considerations
Working with Indian suppliers also means understanding Indian and the importing country’s rules.
Some key points are:
For the US, plants must comply with US FDA cGMP and support ANDA or NDA filings. For the EU, plants must hold EU-GMP certificates, and products must pass assessment by EMA or national agencies.
Across all regions, regulators expect pharmacovigilance systems and fast reporting of side effects and quality issues.
Logistics, Shipping & Cold Chain Management

Once regulatory issues are under control, logistics become the next key success factor.
Temperature-Sensitive Products
For temperature-sensitive products such as vaccines, biologics, and some injectables, maintaining the cold chain is critical.
Use validated insulated shippers or refrigerated containers equipped with temperature loggers to monitor conditions during transport.
Storage and shipping requirements should be clearly defined for example, 2–8 °C, −20 °C, or controlled room temperature and these conditions must be properly aligned with your logistics plan to ensure product stability and quality.
Packaging Validation
Packaging validation is important for export products because packs must withstand long journeys, rough handling, and varying climate conditions.
Transport tests such as vibration, drop, and compression studies should be carried out when required to ensure the packaging can protect the product during transit.
Moisture-barrier materials and desiccants should be used for hygroscopic products to prevent damage from humidity.
In addition, packaging should include tamper-evident features and clear labeling in the local language of the destination market to ensure safety, compliance, and proper identification.
Customs Clearance
Customs clearance requires proper export documentation to ensure smooth shipment processing.
This documentation often includes a commercial invoice and packing list, a Bill of Lading or Air Waybill, and a Certificate of Origin.
In addition, regulatory documents such as the Certificate of Analysis (CoA), GMP Certificate, and any country-specific forms may also be required.
It is important to agree with your supplier in advance about who will prepare each document and at what stage of the shipment process to avoid delays or compliance issues during customs clearance.
Risks & Challenges in Pharma Sourcing
Buying medicines from another country brings real risks that must be managed:

Risk Mitigation Strategies
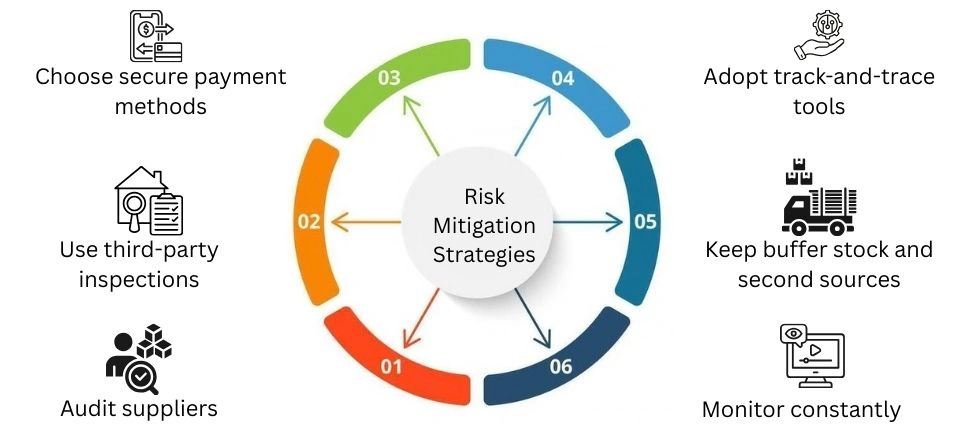
To make the supply chain stronger, buyers can:
Cost Structure & Profit Margins
When you plan your sourcing strategy, you need to see the full cost picture, not just the ex-factory price.
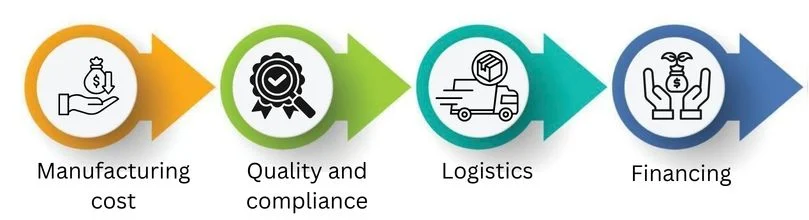
Key cost blocks include:
Because Indian manufacturing is 30-40% cheaper than many Western sites, there is often room to keep good margins even after adding freight and duty.
This is especially true in semi-regulated and emerging markets. In crowded markets like the US or parts of Europe, price pressure is much stronger.
Margins can be thin. Here, you must focus on:
Future Trends in Indian Generic Exports
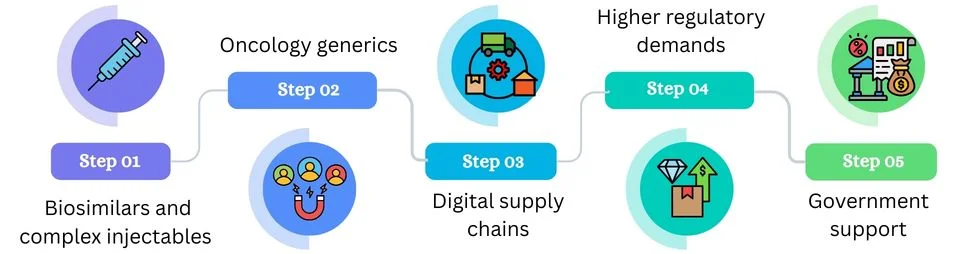
Several trends will shape how Indian generic exports evolve over the next decade:
Why Indian Pharmaceuticals Are in Global Demand
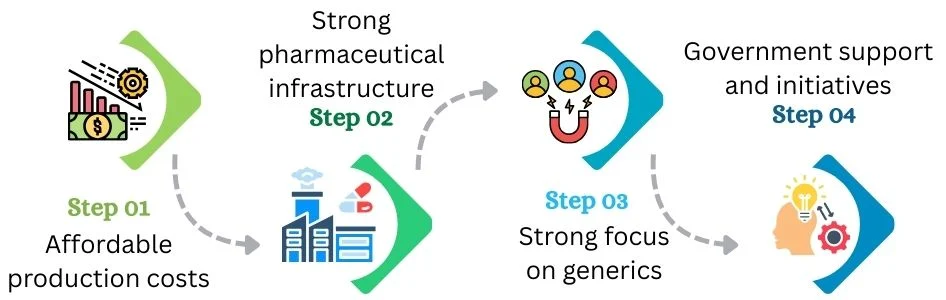
Many structural factors explain why buyers in more than 200 countries rely on Indian medicines:
Conclusion
Sourcing generic medicines from India is now a key choice for many health systems and companies.
It cuts costs while still keeping treatment within reach for a large number of people.
To do this well, buyers need a clear and simple plan.
They must write down what products they need, choose trusted suppliers, check all papers, and test samples in outside labs.
Clear contracts, good shipping plans, and safe cold storage are also very important.
With good planning and careful checks, companies can build strong, long-term links with Indian manufacturers. These links give a steady supply, good profit, and reliable quality.
In the end, this helps both the business and the patients who need low-cost, high-quality generic medicines.
